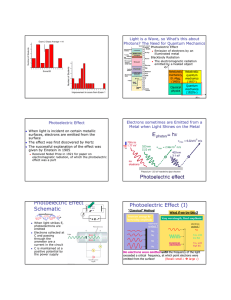
ElectroMagnetic Photoelectric Effect Spectrum
advertisement

ElectroMagnetic Spectrum Quick Quiz: Why do we use infrared waves and not X-rays to heat our homes? A. X-ray wavelengths are too short B. X-ray wavelengths are too long C. We use microwaves to heat our homes D. Physical law says that Xrays can only be used for medicine Discovery: Heinrich Hertz Photoelectric Effect When light is incident on certain metallic surfaces, electrons are emitted from the surface The effect was first discovered by Hertz The successful explanation of the effect was given by Einstein in 1905 For what did Einstein receive the Nobel Prize in 1921? a. Theory of Special Relativity b. Theory of General Relativity c. Theory of Quantum Gravity d. Theory of the photoelectric effect e. Einstein never received the Nobel Prize Hertz’s Spark Gap Generator Back in 1887… Hertz clarified Maxwell’s electromagnetic theory of light: Proved that electricity can be transmitted in electromagnetic waves. Established that light was a form of electromagnetic radiation. First person to broadcast and receive these waves. Electrons sometimes are Emitted from a Metal when Light Shines on the Metal f Classical? Or Quantum? “Classical” Method Increase energy by increasing amplitude electrons emitted ? No No No No What if we try this ? Vary wavelength, fixed amplitude electrons emitted ? No Yes, with low KE Yes, with high KE No electrons were emitted until the frequency of the light exceeded a critical frequency, at which point electrons were emitted from the surface! (Recall: small large f) 1 Photoelectric Effect Schematic I e+V _ When light strikes E, photoelectrons are emitted Electrons collected at C and passing through the ammeter are a current in the circuit C is maintained at a positive potential by the power supply Photoelectric Effect (II) Electrons are attracted to the (positively charged) nucleus by the electrical force In metals, the outermost electrons are not tightly bound, and can be easily “liberated” from the shackles of its atom. It just takes sufficient energy… Classically, we increase the energy of an EM wave by increasing the intensity (e.g. brightness) Meter on Box Energy A2 Digital Meter But this doesn’t work ?? Quantum of Light Energy Enter Mr. Genius! Light is acting like a particle The light particle must have sufficient energy to “free” the electron from the atom. Increasing the Amplitude is simply increasing the number of light particles, but its NOT increasing the energy of each one! However, if the energy of these “light particle” is related to their frequency, this would explain why higher frequency light can knock the electrons out of their atoms, but low frequency light cannot… In this “quantum-mechanical” picture, the energy of the light particle (photon) must overcome the binding energy of the electron to the nucleus. If the energy of the photon exceeds the binding energy, the electron is emitted with a KE = Ephoton – Ebinding. The energy of the photon is given by E=hfwhere the constant h = 6.6x10-34 [J s] is Planck’s constant. “Light particle” After Collision Before Collision Photons Quantum theory describes light as a particle called a photon If Photoelectric Effect were Classical The following graphs illustrate what the wave theory predicts will happen: According to quantum theory, a photon has an energy given by E = hf = hc/ h = 6.6x10-34 [J s] Planck’s constant, after the scientist Max Planck. The energy of the light is proportional to the frequency (inversely proportional to the wavelength) ! The higher the frequency (lower wavelength) the higher the energy of the photon. 10 photons have an energy equal to ten times a single photon. Quantum theory describes experiments to astonishing precision, whereas the classical wave description cannot Icurrent Icurrent Ilight intensity Voltage Icurrent frequency of light 2 Photoelectric Effect is Quantum Effect We now show in green what actually happens: V-stop Ilight intensity Voltage Icurrent f-co In addition, we see a connection between the stopping voltage and frequency above f-co (frequency of cut-off): Quick Quiz If V = hf, the slope of the graph is: V-stop Icurrent Icurrent Stopping Voltage depends on f f-co frequency A. B. C. D. E. 1/h V h f 1/f frequency of light 3