Light is a Wave, so What’s this about
advertisement

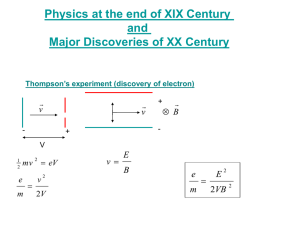
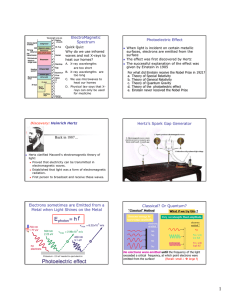
Light is a Wave, so What’s this about Photons? The Need for Quantum Mechanics Exam 2 Class Average = 14 4 3 2 1 5 0 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 Score/20 Number of Students Number of Students 5 4 Photoelectric Effect Emission of electrons by an illuminated metal Blackbody Radiation The electromagnetic radiation emitted by a heated object v/ c Relativistic Relativistic mechanics, quantum El.-Mag. mechanics (1905) (1927-) Quantum Classical mechanics physics (1920’s-) 3 2 1 0 -5 -4 -3 -2 -1 0 1 2 3 4 5 6 7 8 9 10 Improvement in score from Exam 1 h/s Electrons sometimes are Emitted from a Metal when Light Shines on the Metal Photoelectric Effect When light is incident on certain metallic surfaces, electrons are emitted from the surface The effect was first discovered by Hertz The successful explanation of the effect was given by Einstein in 1905 Received Nobel Prize in 1921 for paper on electromagnetic radiation, of which the photoelectric effect was a part Photoelectric Effect Schematic lig ht e- I +V _ When light strikes E, photoelectrons are emitted Electrons collected at C and passing through the ammeter are a current in the circuit C is maintained at a positive potential by the power supply Photoelectric Effect (I) “Classical” Method Increase energy by increasing amplitude electrons emitted ? No No No No electrons emitted ? No Yes, with low KE Yes, with high KE No electrons were emitted until the frequency of the light exceeded a critical frequency, at which point electrons were emitted from the surface! (Recall: small λ large ν)