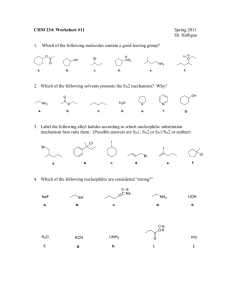
SN1 vs SN2 vs E1 vs E2 Name______________________ Comparison of Reactions:
advertisement

SN1 vs SN2 vs E1 vs E2 Name______________________ Comparison of Reactions: Sn2 vs Sn1 vs E1 vs E2 Substrate Good Nuc, Good Nuc, (RX) Weak Base Strong Base I- Br- HS- OH- RONH3 PH3 NH2methyl SN2 SN2 Species that are Good Nucleophiles but Weak Bases promote SN2 reactions I-, Br-, Cl-, HS-, NH3, PH3, Species that are Good Nucleophiles but Strong Bases promote both SN2 and E2 (depends on substrate) OH-, RO-, H2NSpecies that are Poor Nucleophiles and Weak Bases promote SN1/(E1) reactions H2O, ROH primary SN2 secondary tertiary Predict the Products: CN DMF Cl H b) I- H H OH- I H2O (SN2) (E2) OH d) (SN2) No Rxn No Rxn SN2 Mostly SN2 (some E2) E2 No Rxn E2 SN1 SN1 (SN1) OH- I f) CH3OCl OCH3 g) Cl CH3OH No Reaction + Br (E1) (E2) e) I acetone Cl H2O ROH Predict the Products: CN- Br a) Poor Nuc Weak Base h) H H OCH3 (SN2) OCH3 H CH3OH racemic mixture (SN1) (E1) 1 Sn1 vs Sn2 vs E1 vs E2 Unimolecular Elimination, E1 Name______________________ EI elimination reaction Elimination Reactions: Removal of Leaving Group (L) and H, but no substitution Formation of a C=C double bond Solvolysis of 2-bromo-2-methylpropane With H2O actually gives 2 products (SN1=alcohol, E1=alkene) Rate of reaction for both products K= k[haloalkane] Step 1 is Dissociation (just like for SN1) Bimolecular Elimination, E2 carbocation intermediate 2nd transition state alkene product E2 elimination reaction Strong Bases affect nucleophilic substitution reactions (OH- and RO-) Rate K= [B-][RX] (second order or bimolecular) Must occur before a carbocation forms, or we would just have SN1, E1 E2 Mechanism Occurs in a single, concerted step Must have strong base substrate transition state alkene product 1 Sn1 vs Sn2 vs E1 vs E2 Name______________________ Substitution vs. Elimination Comparison of E1 and E2 SN2, SN1, E2, E1 all occur from RX + Nu E1 is stepwise and involves a carbocation Br CH3 C CH2 H CH2 CH3 + H2O CH3 - R H R + CH3 E2 is concerted HO + H3O C R R R R X R R + OH2 + X How do we predict what the reaction will produce? -Weakly basic, strong nucleophiles give substitution products -Good Nucleophiles -Base weaker than OHSN1 for tertiary RX -I-, Br-, RS-, N3-, RCO2-, PR3, CN-SN2 for primary and secondary RX Weakly basic, weak nucleophiles can only do SN1/E1 (H2O, ROH) Br CH3 C CH3 CH2 H2O CH3 C CH3 Experimental Evidence for Concerted E2 Mechanism: Second order rate law shows base and RX involved in RDS Better L is faster (Bond breaking is important in RDS) Stereochemistry:If anti-H is present, E2 is fast, if not present E2 is Br very slow fast H CH3O slow CH3O - H + OH CH3 Strongly basic Nu’s give more E as Bulk increases (OH-, RO-, NH2-) HO- + Br HO - + Br + Br OH + Br E2 mostly + Br- Br - HO : + Br SN2 mostly - E2 mostly H H Sterically hindered bases favor Elimination -too bulky to do substitution -E2 or E1 depending on RX O- + Br Reactivity of Haloalkanes Primary Haloalkanes SN2 mostly, can be slowed by bulk at βC E2 only if bulky base Secondary Haloalkanes SN2 with good nucleophiles, weak base, Polar Aprotic Solvent SN1/E1 with good L, weak Nu, polar protic solvent E2 with strong base, polar protic solvent Tertiary Haloalkane SN1/E1 with good L, no base (solvolysis) E2 with strong base O- + Br Likely mechanism of reaction of haloalkane with base - X 2