PCR prelab
advertisement

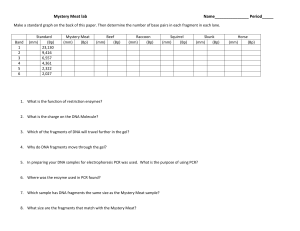
Ex. 26: DNA Amplification of Mycobacterium tuberculosis by PCR is DNA replication in a test tube Objectives ?? Invented by Kary Mullis, 1983. Nobel Prize 1993. “I was working for Cetus, making oligonucleotides. They were heady times. Biotechnology was in flower and one spring night while the California buckeyes were also in flower I came across the polymerase chain reaction. I was driving with Jennifer Barnett to a cabin I had been building in northern California. She and I had worked and lived together for two years. She was an inspiration to me during that time as only a woman with brains, in the bloom of her womanhood, can be. That morning she had no idea what had just happened. I had an inkling. It was the first day of the rest of my life.” - from Karry Mullis’s autobiography at the Nobel e-Museum Did He Really Invent PCR? The basic principle of replicating a piece of DNA using two primers had already been described by Gobind Khorana in 1971: – Kleppe et al. (1971) J. Mol. Biol. 56, 341-346. Progress was limited by primer synthesis and polymerase purification issues. Mullis properly exploited amplification. What is the Polymerase Chain Reaction? • Think of it as a molecular photocopier • It is a means of selectively amplifying a particular segment of DNA. • The segment may represent a small part of a large and complex mixture of DNAs: e.g. a specific exon of a human gene. A Molecular Photocopier A photocopier capable of duplicating a part of a sentence: “The next day was quite a different day. Instead of being hot and sunny, it was cool and misty. Pooh didn’t mind for himself, but when he thought of all the honey the bees wouldn’t be making, a cold misty day always made him feel sorry for them.” A.A. Milne, 1928. The words in blue must be unique for the copier to locate the correct piece of text. How Powerful is PCR? PCR can amplify a single DNA molecule, e.g. from a single sperm. PCR can amplify DNA to a usable amount (visible by gel electrophoresis) in ~2 hours. The template DNA need not be highly purified — a boiled bacterial colony. The PCR product can be digested with restriction enzymes, sequenced or cloned. The Basics of PCR Cycling 30 - 35 cycles, each comprising: – Denaturation (95°C), 30 sec. – Annealing (55–60°C), 30 sec. – Extension (72°C), time depends on product size. DolanDNA Learning Center: PCR Tutorial DolanDNA Learning Center on YouTube Additional narrated PCR Tutorial What’s in the Reaction? 1. Template DNA 2. Reaction buffer and magnesium ions 3. Nucleotides (dNTPs) 4. Primers 5. DNA polymerase (usually Taq) So Then, it’s Easy? Cycling performed with three water baths. Thermal cyclers introduced in 1986. Early polymerases were not thermostable, so had to be replenished each cycle. The 37°C temperature caused non-specific priming, resulting in unwanted products. Taq (Thermus aquaticus) DNA polymerase first described in 1988. Forensic Microbiology • Primer for a specific organism will allow for detection that particular organism RT-PCR with a norovirus primer • Real-time PCR: Newly made DNA tagged with a fluorescent dye; the levels of fluorescence can be measured after every PCR cycle • Reverse-transcription PCR (RT-PCR): Reverse transcriptase makes DNA from viral RNA or mRNA Textbook Clinical Focus, p. 266 Day 1 Materials needed per 2 tables: Four 200 µl PCR tubes containing PCR beads Micropipette and tips capable of measuring out 10 µl Patient samples Positive control sample Distilled water for negative control Equipment: Nano centrifuge, PCR Thermocycler Work in a team of 8 students. Part 2 of Day 1 Cast, Load, and Electrophorese a 1.5 % Agarose Gel Materials needed per table: Amplified PCR products from last session Bottle of 0.5 X TBE (Trisborate/EDTA) buffer 50 ml measuring cylinder, 250ml Erlenmeyer flask Minigel electrophoresis apparatus with gel tray, spacers, and comb micropipettes and matching tips Microcentrifuge tube containing 20 µl of 10x gel loading buffer. Materials shared by whole class: 1. Scales, flasks, electrophoresis grade agarose, cylinder, and TBE buffer at “gel making station” 2. Microwave 3. Power source (4 available per class) 4. Nanofuge (2 per room) 5. DNA standard (instructor handles it) 6. “InstaStain” ethidium bromide staining sheets (handled or supervised by the instructor). 7. Gel documentation system Understanding Gel Electrophoresis Make 30 ml of a 1.5% agarose gel. How? How does gel electrophoresis separate DNA fragments? • Gel acts as sieve to filter DNA fragments • DNA fragments are naturally negatively charged (phosphate backbone) • DNA pulled towards anode (pos. electrode) by electric attraction • Smaller fragments move faster through the gel and larger fragments move slower • gel electrophoresis is optimized by adjusting agarose concentration in gel + + + + + + + + + + + + + + + + + + + + + + + + ++ + + + + + + + + + DNA is negatively charged and therefore repelled from the negative pole and attracted towards the positive pole --------------- ------ --- --- --- ---- - + + + + + + + + + + + + + _ _ _ _ _ _ _ _ _ _ _ _ A Typical Image of an Agarose Gel Under UV Light Decreasing DNA Size Largest DNA fragments Smallest DNA fragments The Intensity of the Band is Proportional to the Concentration Of DNA • An important point to remember is that the intensity of the band is proportional to the amount of DNA found in the band The upper band has far less DNA when compared to the lower band. The intensity of the bands are proportional to the amount of DNA at that position in the gel Sizing The Fragments of DNA The sizes of the various fragments can be identified by including a “ladder” in the gel – A ladder is a mixture of DNA fragments of known size – A ladder is usually run beside the unknown sample so that the sizes of the various DNA fragments in the sample can be identified Marker / Ladder / Size Standard • Mixture of DNA fragments of selected sizes • When run in a gel, fragments will separate into distinct bands that can be used as references • Fragment size always stated as [X] base pairs (bp) • Two common ladders: 200bp and 1kbp (1000 bp) ladders • 200 bp ladder composed of a mixture of small fragments (200 to 4000 bp) • Ladders commercially available Sizing a Gel Product Base Pairs (bp) 4000 3000 2000 1600 2000 bp 1000 bp 1000 4000 3000 500 2000 1600 1Kbp Sample 1000 ladder 500 Size of this Fragment? Sample 1Kbp ladder The size of the fragment is…?? Size In Base Pair (bp) 1500 1200 1000 600 500 400 300 200 100 1 kbp ladder was used ? 4000 3000 2000 1600 1000 500 Music video from "Scientists for Better PCR" and Bio-Rad.