polymers - Liberty Union High School District
advertisement

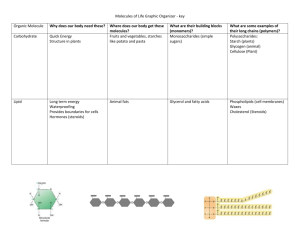
Biochemistry and Polymers Biological Macromolecules and Polymers Standards 10. The bonding characteristics of carbon allow the formation of many different organic molecules of varied sizes, shapes, and chemical properties and provide the biochemical basis of life. As a basis for understanding this concept: a. Students know large molecules (polymers), such as proteins, nucleic acids, and starch, are formed by repetitive combinations of simple subunits. 10. b. Students know the bonding characteristics of carbon that result in the formation of a large variety of structures ranging from simple hydrocarbons to complex polymers and biological molecules. 10. c. Students know amino acids are the building blocks of proteins. 10. f.* Students know the R-group structure of amino acids and know how they combine to form the polypeptide backbone structure of proteins. Biological Molecules • Most biological chemical compounds are molecules – they have covalent bonds (because they are mostly made up of the nonmetals: H, C, O, and N). • Remember: Nonmetal with Nonmetal - form covalent compounds • macromolecules – large molecules, usually from biology. Polymers • polymers – long chain molecules that have a repeating monomer unit. means many Examples: –A–A–A–A–A–A– monomer unit O H O H O H C C O H C C O H C C O H C C O H C C O H H H O O Polymers Continued • Carbon atoms usually form the “backbone” of polymers because carbon atoms can form: C C C C C C Single bonds Double bonds Triple bonds These allow the polymers to have various structures and keep on going … Biological Polymers • Proteins are polymers made up of amino acids chained together. • Cellulose and starches are polymers made up of sugars chained together. • Most fats and oils are polymers made up of fatty acids chained together. • Deoxyribonucleic Acid (DNA) is a polymer made up of sugar phosphates chained together with nucleotides in the middle. H He Li Be B C N O F Na Mg Al Si P S Cl Ar K Ca Ne Br Kr I Xe H He Li Be B C N O F Na Mg Al Si P S Cl Ar K Ca Ne Br Kr I Xe 4 e– in valence shell