L3t3
advertisement

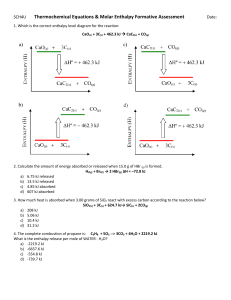
Whole class detention on Friday because of those who didn’t do homework REVIEW COMBUSTION • ______________ is a process in which a selfsustaining chemical reaction occurs at temperatures above those of the surroundings. • EXOTHERMIC = RELEASES HEAT • ENDOTHERMIC = ABSORBS HEAT • ________________ = is a measure of the total ENTHALPHY energy possessed by a substance or group of substances. REVIEW • What is the equation for ∆H ? ΔH = enthalpy of products – enthalpy of reactants • Is the following reactions endothermic or exothermic? EXPLAIN – – – – 2SO2( g) + O2( g) → 2SO3( g) CH4( g) + H2O( g) → CO( g) + 3H2( g) NH3(aq) + HCl(aq) → NH4 +(aq) + Cl–(aq) Ag+(aq) + I–(aq) → AgI(s) ∆ H = –198 kJ/mol ∆ H = +206 kJ/mol ∆ H = –52 kJ/mol ∆H = –112 kJ/mol REVIEW • Is the following reaction endothermic or exothermic? 2N + 6H N2 + 3H2 2NH3 • What formula for ∆H would you use in this circumstance? ∆ H = (energy to break reactants) – (energy to make product) TEMPERATURE • Temperature is a measure of the degree of hotness or coldness of an object or substance. The hotter the object, the higher its temperature. • If two objects or samples of material are brought into contact, heat will flow from the hot object to the cold one until the temperature of the two objects is equal. • When the temperature is uniform throughout both objects or samples of material, we say that thermal equilibrium has been reached. Quantity of Heat • Two objects can be at the same temperature but contain very different amounts of heat. EXPERIMENT 1 • Three well-insulated beakers each contain 100 g of water at 25.0°C. The quantity of heat depends upon mass, and nature of substance, Factors Effecting Quantity of HEAT • The amount of heat energy is proportional to the mass of the substance involved. • The amount of heat energy contained in equal masses of different substances depends upon the nature of the substances involved. SPECIFIC HEAT CAPACITY Units = joules per kelvin per gram, (J/Kg) (J K-1 g-1). Calculating Quantities of Heat Specific heat capacities allow us to calculate the quantities of heat that flow from one object or substance to another. Where: q = quantity of heat involved m = mass of substance C = specific heat capacity ∆ T = change in temperature (Final – Initial) When q is positive temperature increase When q is negative temperature decreases HOMEWORK Check