Atomic Structure
advertisement

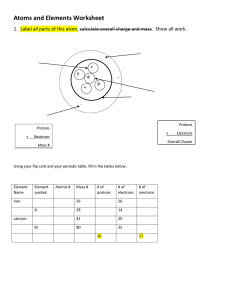
The Structure of the Atom Chemistry 1 Learning Objectives for this Chapter: 1. Describe changes in the atomic model over time and why those changes were necessitated by experimental evidence. 2. Describe structure of atoms (protons, neutrons, electrons) and differentiate among these particles in terms of mass, charges and location in the atom. 3. Compare the magnitude and range of the 4 fundamental forces. 4. Apply the mole concept and the Law of Conservation of Mass. 2 It started a long time ago… Today 460 – 370 BC Democritus Beginning of Atomism You cannot divide something in half forever. The smallest piece of matter is called an atom. 3 Dalton’s Postulates 460 – 370 BC 1808 Democritus Atomism Today 1. All elements are made of tiny indivisible particles called atoms. 2. All atoms of the same element are the Over 2,000 years later John Dalton comes up with the first “modern” atomic theory. same, but different from atoms of every other element. 3. Chemical reactions rearrange atoms but do not create, destroy, or convert atoms from one element to another. 4. Compounds are made from combining atoms in simple whole number ratios. 4 Another kind of light? 460 – 370 BC 1808 Democritus Atomism Dalton “Modern” atomic theory 1870 Today William Crookes invents a tube in which virtually all the gas has been removed. Under high voltage, a ray was emitted from the cathode end of the tube. 5 It started a long time ago… 460 – 370 BC 1808 Democritus Atomism Dalton “Modern” atomic theory 1870 Crookes Cathode rays Cathode rays must be negative. 1897 Today J.J. Thomson discovers the electron 6 It started a long time ago… 460 – 370 BC 1808 Democritus Atomism Dalton “Modern” atomic theory 1870 Crookes Cathode rays 1897 1910 Thomson Discovery of the electron Today Ernest Rutherford discovers the nucleus 7 Size and mass The size of the atom comes mostly from the space occupied by the electrons The mass of the atom comes mostly from the nucleus 8 neutrons electrons protons What happens when you change the number of protons? 9 6 protons in carbon 7 protons in nitrogen 8 protons in oxygen You obtain a different element! The number of protons is also called the atomic number for that element. 10 neutrons electrons protons What happens when you change the number of electrons? You get an ion – a charged particle. 11 A neutral sodium atom 11 protons Na The protons and electrons cancel each other out Balanced charges A positive sodium ion 1+ Na One proton is not neutralized by an electron, making this a +1 charged atom One electron short 12 8 protons A negative oxygen ion -2 O Two electrons are not neutralized by protons, making this a –2 charged atom Two extra electrons 13 The electron cloud Electrons are very light and fast. They are not organized along orbits around the nucleus. Except for mass, virtually every property of atoms is determined by electrons, including size and chemical bonding 14 neutrons electrons protons What happens when you change the number of neutrons? 15 Atomic number Neutrons act as “glue.” They hold protons together in the nucleus. 16 Mass number = 6 p + 6 n = 12 12C “carbon-12” mass number: total number of protons and neutrons in a nucleus. 17 Name Mass number # protons # neutrons Carbon-12 Carbon-13 Carbon-14 12 13 14 6 6 6 6 7 8 isotopes: atoms or elements that have the same number of protons in the nucleus but different number of neutrons 18 Atomic particle determination a. Neutrons = mass number of nucleotide – atomic number of element b. Protons = atomic number (Z) of element c. Electrons = atomic number of element d. Atomic Mass – average mass of isotopes of an element e. Mass number – the total protons and neutrons in the nucleus of an atom 19 V Atomic Mass Formulae 1. Mole – the amount of a substance that contains the same number of units as the number of atoms in exactly 12 g of Carbon – 16. This is Avogadro’s number of 6.022 x 1023 particles. 2. Mass to number of atoms = given mass in g of element 6.022 x 1023 atoms Molar mass in g of element 3. Number of atoms to mole: # atoms mole 6.022 x 1023 atoms 4. Mass to moles: given mass in g moles Molar mass in g 20 Number of neutrons for each of 100 lithium atoms randomly sampled from nature It’s an AVERAGE mass! 21 Isotope periodic table (first 4 rows) 22 Forces found in Nature: 1. Gravitational – relatively very weak but active over long distances 2. Electromagnetic – intermediate strength 3. Strong Nuclear – strongest force that binds protons and neutrons together in the nucleus 4. Weak Nuclear – involved in the decay of many elementary particles 23 Examples of the Weak Nuclear force. Some atoms are unstable and, to become stable, decay by emitting particles or energy or both. Alpha particles, a Beta, b 24 Nuclear Particles Proton Mass: 1 amu Charge: +1 Neutron Mass: 1 amu Charge: 0 (1 amu = 1/12 mass of Carbon 12 atom) 25 Electrons Negative 0 e -1 Positive 0 e positron +1 26 Uranium Isotopes 238 235 U 92 U 92 Low Radioactive Fission 27 Nuclear Reactions • Nuclear Decay - a spontaneous process in which an unstable nucleus ejects a particle and changes to another nucleus. – Alpha decay – Beta decay • Beta Minus • Positron • Fission - a nucleus splits into two fragments of roughly equal size • Fusion - Two nuclei combine to form a heavier nucleus. 28 Alpha Decay • This occurs when a helium nucleus is released. • This occurs only with very heavy elements. 29 Beta (b-) Decay • A beta particle (negative electron) is released when a nucleus has too many neutrons for the protons present. A neutron converts to a proton and electron leaving a greater number of protons. • Neutron decay: 30 Positron (b+) Decay • Positron decay occurs when a nucleus has too many protons for the neutrons present. A proton converts to a neutron. A neutrino is also released. 31 Fission Fusion 32 Learning Objectives for this Chapter: Atomic Structure 1. Describe changes in the atomic model over time and why those changes were necessitated by experimental evidence. 2. Describe structure of atoms (protons, neutrons, electrons) and differentiate among these particles in terms of mass, charges and location in the atom. 3. Compare the magnitude and range of the 4 fundamental forces. 4. Apply the mole concept and the Law of Conservation of Mass to make calculations. 33