Aspirin_Resistance
advertisement

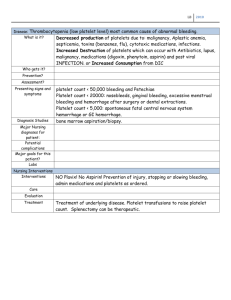
Aspirin Resistance Clinical Questions How do we define aspirin resistance? Is this concept clinically relevant? Why aren’t we testing for it? Aspirin Basics Antithrombotic Trialists’ Collaboration Major meta-analysis reporting 25% reduction in significant vascular events as secondary prevention Prevents at least 10-20 fatal and nonfatal vascular events for every 1000 patients treated for one year. American Heart Association recommends prophylaxis for: 10yr risk of CHD >10% Pharmacology Consistent absorption via passive diffusion in GI tract with peak plasma levels in 30min Enteric coated peak in 3-4 hours with variable bioavailability Platelet Basics Wide acceptance that platelet activity plays a major role in atherothrombosis Adhesion: Platelet membrane receptors bind to endothelium and subendothelium at sites of damage Activation: Transmembrane signaling increases surface receptors, granule release, and exposure of membrane phospholipid Aggregation: Procoagulant surface of the membrane serves as basis for coagulation cascade, amplification of platelet response, and production of fibrin. Aspirin Inhibition of Platelets Aspirin inhibits platelet cyclooxygenase (COX)-1 thereby preventing formation of thromboxane A2 (TxA2), a potent aggregating and vasocontrictor agent. Acetylation serine 529 of COX-1 Platelets – anucleate and unable to regenerate COX-1 • Platelet lifespan 7-10 days • Maturation time from megakaryocytes: 4 days NSAID Mechanism Definition of “Aspirin Resistance” Clinical failure of prevention Biochemical resistance Laboratory Phenomenon Or…. Patient just doesn’t want to take it. Clinical “Aspirin Resistance” Perhaps more appropriate term would be “treatment failure” Probabilities Game Failure is to be expected in the treatment/prevention of any multifactorial disease process Theories of Biochemical Resistance Endothelial cells recover COX-1 activity shortly after aspirin exposure Role of COX-2 in thrombotic process Thromboxane-independent methods of platelet activation Genetic polymorphisms GP IIb-IIIa receptor Platelet alloantigen 2 (PlA2) Laboratory Definition Bleeding time Aggregometry Platelet function analyzer (PFA-100) Platelet Release Products VerifyNow Aspirin Platelet Membrane Expression Bleeding Time Should not be used Low Sensitivity Invasive Poorly reproducible Multiple Variables • • • • • Platelet function Platelet count Plasma factors Red Blood Cells Skin trophism Aggregometry Light Transmission Aggregometry Measures light transmission through a platelet suspension in response to agonist (arachodonic acid or ADP) Reports: % residual platelet function Clinical association with vascular events Historic Reference Standard time-consuming and user-dependent limited availability and requires rapid analysis lack of standardization Aggregometry Whole Blood Aggegrometry Measures electrical impedance between two electrodes immersed in whole blood 5min after addition of a platelet agonist Reports: impedance in Ω Easy, sensitive, reproducible Large sample of blood required Long preparation time Expensive Platelet Function Analyzer-100 Anticoagulated blood is aspirated through a capillary and a 150μm aperture coated with collagen and ADP or epinephrine. Measures time needed until blood flow interruption is recorded. Reports: closure time (<193s) No association with vascular events Point-of-Care test Must test within 4hrs of blood collection Dependent on hematocrit and vWF Platelet Release Products Serum TXA2 Urinary 11-dehydrothromboxane B2 Directly dependent on aspirin’s effect on COX-1 inhibition Simple and correlated with clinical events Indirect measurement Not specific to platelets Urinary test dependent on renal function VerfiyNow Aspirin Turbidometric-based optical system that measures agglutination of fibrinogen-coated beads by platelets stimulated in citrated whole blood. Reports: Aspirin Reaction Units Clinical association with vascular events Point-of-care test Major limitation relates to the diagnostic criteria set for definition of aspirin resistance Compared to optical aggregometry with epinephrine after one 325mg dose of aspirin Platelet Membrane Expression Flow cytometry allows for the evaluation of platelet reactivity and in vivo activation. However, these techniques require sophisticated and expensive instruments. Platelet bound P-selectin Soluble plasma P-selectin Platelet-leukocyte aggregates Platelet-derived microparticles A comparison of six major platelet function tests to determine the prevalence of aspirin resistance in patients with stable coronary artery disease. Marie Lordkipanidze, Chantal Pharand, Erick Shampaert, Jacques Turgeon, Donald Palisaitis, and Jean G. Diodati. European Heart Journal in June 2007 Study Design 201 patients with stable CAD on daily aspirin therapy (>80mg) Consecutive patients diagnosed with CAD Montreal, Canada Sample collection: morning urine and blood Exclusions: ACS or revascularization within 6 months Concurrent use of NSAIDs, clopidogrel, dipyridamole, warfarin, acenocoumarol Major surgery within 1 month of enrollment Thrombocytopenia, Hct <25% Dialysis Platelet Function Tests LTA with arachidonic acid LTA after adenosine diphosphate Whole blood aggregometry PFA-100 VerifyNow Aspirin Urinary 11-dehydrothrombaxane B2 Patient Data 155 Male (71%) Mean Age: 66.5 ± 10.4 All with aspirin ≥ 1 month 80mg – 110 patients 81mg – 10 patients 162.5mg – 1 patient 325mg – 79 patients 1300mg – 1 patient Results κ Statistic 0 - 0.2 0.2 - 0.4 0.4 - 0.6 0.6 - 0.8 Agreement Slight Fair Moderate Substantial Prevalence of Resistance Discussion Overall, poor correlation between different platelet function tests “Aspirin resistance” based on arbitrary, clinically non-validated cut-off values. “We believe the non-standardized use of these assays and the absence of a formal definition explains much of the disparity reported in the literature in regards to the prevalance of aspirin resistance.” Perhaps it is more important to determine cutoff values with the intent of predicting clinical outcomes instead of focusing on a “gold standard” test. Clinical Confounders Drug Interactions: NSAIDs Comorbid Conditions Isoprostanes may occupy nearby catalytic site Block access of aspirin to serine 529 Ibuprofen, Indomethacin PPIs? ACE Inhibitors? prostaglandin F2-like compounds Produced from arachidonic acid in a nonCOX process Amplify platelet response Smoking Diabetes Hyperlipidemia, Unstable Angina Variable Aspirin Response Most studies have not evaluated aspirin resistance over time Cohort study out of the University of Chicago published in 1994 Secondary prevention in 306 patients with history of ischemic stroke Repeated assessments of aspirin resistance over a 6 month interval Light Transmission Aggregometry 32.7% of patients did not maintain complete inhibition with repeated testing Reassessment of fixed-dose regimens? Patient Compliance No laboratory testing to monitor for clinical adherence Schwartz, et al. studied tested 190 patients using LTA and found 9% resistance Repeat testing after directly observed therapy in all 9% Nonresponders responders Tantry, et al. studied 223 patients with CHD All but 7 patients had aspirin response via LTA Those 7 patients were interviewed again and admitted to noncompliance Working Group on Aspirin Resistance in 2005 “Other than in research trials, it is not currently appropriate to test for aspirin ‘resistance’ in patients or change therapy based on these tests.” Summary “Aspirin Resistance,” from biochemical and lab perspective, probably has clinical relevance There continues to be significant investigation and discovery regarding aspirin therapy Clinical application is on the distant horizon. Consider alternatives to NSAIDs Focus on patient compliance seek to dispel myths assess for and control side effects. References Airee, et al. Aspirin resistance: disparities and clinical implications. Pharmacotherapy 2008; 28(9): 999-1018. Antithrombotic Trialists Collaboration. Collaborative metaanalysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 2002; 324: 71-86. Cattaneo, M. Resistanc to antiplatelet drugs: molecular mechanisms and laboratory detection. J Thromb Haemost 2007; 5 (Suppl 1): 230-7. Christiaens, et al. Major clinical vascular events and aspirinresistance status determined by the PFA-100 method among patients with stable coronary artery disease: a prospective study. Blood Coag and Fib 2008. 19(3): 235-9. Gasparyan, et al. The role of aspirin in cardiovascular prevention. J Am Coll Cardiol 2008. 51; 19: 1829-43. Gum, et al. A prospective, blinded determination of the natural history of aspirin resistance among stable patients with cardiovascular disease. J Am Coll Cardiol 2003; 41(6): 961-5. Helgason, et al. Development of aspirin resistance in persons with previous ischemic stroke. Stroke 1994. 25: 2331-2336 Lordkipanidze, et al. A comparison of six major platelet function tests to determine the prevalence of aspirin resistance in patients with stable coronary artery disease. Euro Heart J 2007; 28(14): 1673-5. MacDonald, et al. Effect of ibuprofen on the cardioprotective effect of aspirin. Lancet 2003. 15; 361(9357): 573-4. Michelson, et al. Aspirin resistance: position paper of the Working Group on Aspirin Resistance. J Thromb Haemost 2005; 3: 1309-11. Patrono, et al. Low-dose aspirin for prevention of atherothrombosis. N Engl J Med 2005. 353; 22: 2373-83. Sanderson, et al. Narrative review: aspirin resistance and its clinical implications. Annals of Int Med 2005. 142; 5: 370-80. Viola et al. Aspirin resistance: is this term meaningful? Curr Opin Hematol 2006. 13: 331-336.