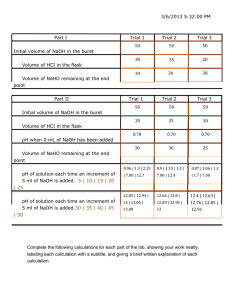
Chemical Analysis by Redox Titration Purpose: Titration is a
advertisement

Chemical Analysis by Redox Titration Purpose: Titration is a common method for determining the amount or concentration of an unknown substance. This is particularly well suited for acid-base reactions and oxidation-reduction reactions. In this experiment you will conduct redox titrations using a standardized permanganate solution (from potassium permanganate). You will be trying to find the % hydrogen peroxide in a commercially sold solution. Background: Permanganate ion is a powerful oxidizing agent, especially in acidic solution, which can be used to analyze solutions. In these titration reactions, the intensely colored permanganate ion is reduced to form the colorless manganese ion. An advantage to using this is that it is “self0indicating”. As long as the reducing agent remains present in the sample, the color of permanganate quickly disappears as it is reduced. However, at the endpoint, all the reducing agent has been used up so the next drop of permanganate solution is sufficient to cause a visible color change. So at the endpoint, we know the oxidizing agent and reducing agent have reacted exactly in proportion to their stoichiometry in the balanced redox reaction. If we know how much of the oxidizing agent we added, then we can figure out how much reducing agent was present in the unknown. Prelab Questions: 1. Identify the hazards of the materials involved in lab. 2. Write the balanced redox equation for this reaction? Show what is being oxidized and what is being reduced. 3. Show the two half reactions and label them. 4. How many electrons are being transferred in each of these half reactions? 5. What color does the permanganate ion characteristically show? 6. What is a titrant? 7. What is hydrogen peroxide used for? Titration Notes: 1. Always rinse the buret with water first then a small about of the titrant. 2. Read the buret carefully (bottom of meniscus). You should be able to estimate to the hundredths place. 3. Do not waste time refilling it to zero for each titration. However by sure to record the volume at which you start every time. 4. Learn to swirl the flask without removing it from underneath the buret. 5. Use a drop, drop, drop pace until you see the color change beginning. Then proceed drop wise. 6. Second and third trials should go much quicker assuming your sample is the same. If the first took 25 mL then you can add 22 mL all at once and then proceed by drops carefully. 7. Diluting with water does not affect “how many” sample molecules are in flask, merely helps you see color change better. 8. Always rinse the sample flask before each titration. 9. Be sure to label everything – including burets and flask. Materials Buret, buret clamp, ringstand 250 mL Erlenmeyer flask 0.100 M KMnO4 solution 3.0 M H2SO4 commercial hydrogen peroxide Procedure: 1. Obtain approx.. 10.0 grams of commercial hydrogen peroxide (record mass accurately). Transfer to clean 250 mL erlenmeyer flask. Rinse the peroxide container twice with a small amount of DI water and transfer to the flask. Dilute the sample flask to about 75 mL with water, then add about 20 mL of 3.0M H2SO4. 2. Set up the buret. Remember to rinse with water then rinse with a small amount (3-5 mL) of the standard KMnO4 solution. Fill the buret with standard solution. Record your initial (starting) volume. 3. Now slowly begin titrating the peroxide solution while continusously swirling the flask. Continue until you see the color become more presistant, at which point you should slow down to dropwise. Continue until one drop produces a color that lasts for at least 30 seconds. Record the final buret reading. 4. Complete a second (and third) trial if time permits. Data: For each trial should record: Mass of peroxide Concentration of standard solution Initial and final buret readings Volume of permanganate used observations Calculations: 1. Determine the moles of KMnO4 solution needed to react. 2. Determine mass and moles of peroxide in each trial. 3. Determine the %H2O2 by mass for each trial. 4. Calculate the average %H2O2. Questions: 1. What indicated the oxidation-reduction was complete? 2. What should have been the limiting reactant in this lab? Excess? How do you know? 3. Why would different %H2O2 be important? 4. Analyze your data, were your trials precise? 5. Compare your sample to those of the class. Is your % higher? Lower? Does age of the material make a difference?