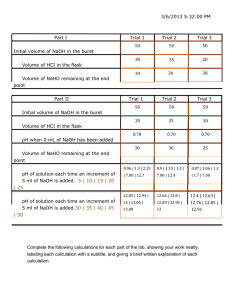
Background Information (Titration) Lab III (iv) Titration is used in
advertisement

Background Information (Titration) Lab III (iv) Titration is used in volumetric analysis of an unknown concentration of a solution. Titration involves adding a solution (the titrant) from a buret to another solution (the sample) in an Erlenmeyer flask until a recognizable endpoint, such as a color change, occurs. PURPOSE: Determine the titration curve of a titration between sodium hydroxide and hydrochloric acid MATERIALS: - 0.5 M hydrochloric acid - 0.1 M sodium hydroxide - buret - ring stand assembly - 125 mL Erlenmeyer Flask - universal indicator solution in dropper bottle - 10 mL graduated cylinder - distilled water in bottle - small 100 mL beaker PROCEDURE: 1. Collect approximately 60 mL of NaOH using the 100 mL beaker. Rinse the buret with small volumes of distilled water using a wash bottle. Using a buret funnel, rinse with small volumes of the NaOH. 2. Using a small funnel, pour the NaOH into the buret until the level is near the top. Open the stopcock for maximum flow to clear any air bubbles from the tip and to bring the liquid level down to the scale. 3. Record the initial buret reading to the nearest 0.1 mL. Avoid parallax errors by reading volumes at eye level. A meniscus finder or placing a piece of paper behind the buret will aid the reading. Volume readings are taken at the bottom of the meniscus. 4. Using the graduated cylinder, measure out 10 mL of HCl and pour it into the Erlenmeyer flask. Place a white piece of paper beneath the flask to make it easier to detect color changes. 5. Add 5 drops of the universal indicator IN THE FLASK. Indicators change color when a specified pH is reached and can be useful in detecting the endpoints of acid-base reactions. Often, a few drops of the indicator will be sufficient to produce a noticeable color change at the endpoint. The indicator, phenolphthalein changes from colorless to pink at approximately pH = 8 and therefore it is commonly used in titrations of acids with strong bases. 6. Open the stopcock so that the titrant is added drop by drop. As the titrant is added, swirl the Erlenmeyer flask gently. At each point that the colour appears to change permanently, stop close the biruet to stop the addition of the titrant. 7. Record the final buret reading to the nearest 0.1 mL. 8. Continue adding titrant until you get your next colour change. 9. Repeat this two more times. 10. When titration is complete, drain the buret into a waste beaker and rinse the buret several times using tap water and finally distilled water. Dispose of the contents of the waste beaker as directed. OBSERVATIONS: Trial 1 Trial 2 Trial 3 i. Volume for Red pH 3 ii. Volume for orange pH 4 iii. Volume for yellow pH 6 iv. Volume for green pH 7 v. Volume for blue pH 10 ANALYSIS: 1) Graph each of the Trial’s data putting pH on the y-axis and the volume added on the x-axis. 2) What are some potential sources of error in this lab? 3) Why do you suppose pH changes over such a narrow range? 4) a. Describe the change in the concentration of hydrogen ions from start to finish. b. Describe the change in the concentration of hydroxide ions from start to finish. Bonus: HCl releases one hydrogen ion in solution. What would a titration graph for an acid like H3PO4 which releases three hydrogen ions in solution look like. Sketch it out.