chemical composition.
advertisement

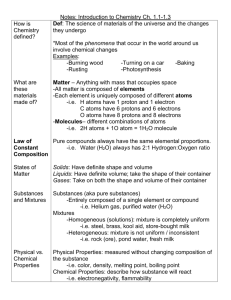
Conceptual Chemistry UNIT 2 – MATTER AND CHANGE Objective 1 Read and express information given in chemical formulas. What is matter? Matter: Anything that has mass and takes up space. Pure Substances Matter that has fixed composition and properties. Cannot be broken down by physical methods. Elements A substance that cannot be broken down by chemical means. i.e. iron (Fe), carbon (C), oxygen (O), nitrogen (N) Compounds A substance made of elements that are chemically combined. i.e. water (H2O), salt (NaCl), sugar (C12H22O11) Compounds Every compound is composed of the elements it contains. Chemical Formula Shows the type and number of atoms present in a substance. Chemical Formula • In the chemical formula, subscripts show how many atoms are in the compound. C6H10N2O2 C=6 H= 10 N= 2 O= 2 Chemical Formula The large numbers in front of the chemical formula, called coefficients, show the number of particles. 3C6H10N2O2 C6H10N2O2 = 3 total Practice Problems: How atoms of each element are in the following? 1. CH4 2. AlCl3 3. 4 H2O 4. 10 Fe3P2 5. 6 Zn(NO3)2 6. Al2(SO3)3 Objective 2 Classify matter as a pure substance or a mixture. Mixtures Matter that has compositions that are not uniform or definite. Mixtures Mixtures can be separated by physical means into the substances that are mixed with in them. Homogeneous Mixtures Homo = same The composition is the same throughout Examples Solid Solid: Brass (Zinc and Copper) Liquid Liquid: Alcohol (Ethanol and Water) Gas Gas: Air (Nitrogen and Oxygen) Solid (dissolves) Liquid: Salt Water (Sodium Chloride and Water) Gas (dissolves) Liquid: Carbonation (Carbon Dioxide and Water) Heterogeneous Mixture Hetero = different The composition is different throughout. Objective 3 Distinguish between chemical and physical changes, as well as chemical and physical properties. Practice Problems Hetero or Homo Mixture Tap Water :_______________ A Rock: ________________ Stainless Steel:________________ Brewed Coffee: ________________ Sand: ________________ Oatmeal: ________________ Dust Free Air: ________________ Rusty Metal: _________________ Why? Properties of Matter Physical Properties: Can be measured without changing the substance’s chemical composition. i.e. melting point, boiling point, density, solubility, odor, hardness, color, etc. Chemical Properties: Potential reactivity of substance with other substances. Can only be measured by trying the reactions. i.e. Any particular reaction a substance undergoes. Changes of Matter Physical Change Chemical Change Physical Change Alters a substance without changing the substance’s chemical composition. i.e. melting, boiling, cutting, smashing, dissolving Chemical Change Occur when one or more substances are changed into another substance that have different chemical compositions. i.e. rusting, exploding, decomposing, corroding, spoiling Practice Problems Chemical or Physical Property? Chemical or Physical Change? Tarnishes in Rain: Lighting a firework: Bends easily: Boiling Pasta: Decomposes: Chewing gum: Reusable/Recyclable: Running your car: Shiny: Rolling your bike: Not Flammable: Melting a crayon: Perishable Food: Rusting a nail: Freezable Food: Opening a can of pop: Objective 4 Balancing chemical equations. Chemical Equation Ex: 2 KClO3 2 KCl + 3 O2 Describes the original beginning substances (called reactants) using chemical formulas. Then an arrow points to the newly created substances (called products) using chemical formulas again. Reactants Products Symbols (s) = += (l) = = (g) = ∆= (aq) = H2 (subscripts) = 2 H2 = (Coefficients) = Balancing Equations The law of conservation of mass states that matter cannot be created or destroyed. So, the number of atoms reacting must equal the number of atoms produced. However, a chemical change rearranges these atoms into new substances. To balance equations, follow these steps: Count atoms on each side of the arrow. They should be equal. If not, you need to balance them. 2. Balance both sides by using coefficients. Remember, they multiply through everything inside of that compound. NEVER change subscripts. 3. Double check work. 1. Practice Problems ____ KClO3 ____ KCl + ____ O2 ____AlBr3 + ____ K2SO4 ____ KBr + ____ Al2(SO4)3 ____ C3H8 + ____ O2 ____ CO2 + ____ H2O Objective 5 Describing chemical reactions and calculate percent yield and percent error. Error Calculations Error = experimental value – accepted value Magnitude of the value tells whether or not the experimental value is too high or too low Accepted value: Accurate value based on a reliable reference Experimental value: value measured in lab Percent Error = | Error | x 100% Accepted Value Percent Yield Used to determine completeness of a reaction Actual Yield: actual amount of products produced in an experiment Theoretical Yield: maximum amount of products predicted from conservation of mass Percent Yield = Actual Yield x 100% Theoretical Yield