Assessment on Formula and
advertisement

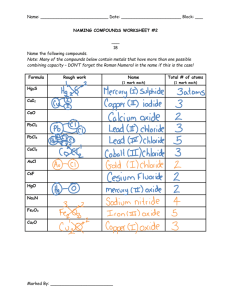
Assessment on Formula and Calculations Core 1. What does the chemical formula tell us about the compound? (1) 2. Write the formula for the following compounds (a) Sodium Chloride (b) Potassium sulphide (c) Aluminium oxide (d) Hydrogen sulphide (4) 3. Write the formula for the following compounds (a) Carbon monoxide (b) Sulphur dioxide (c) Carbon tetrachloride (d) Dinitrogen tetroxide (4) 4. Write the formula for the following compounds (a) Copper (I) oxide (b) Silver (I) chloride (c) Iron (III) sulphide (d) Cobalt Chloride (4) 5. Write the formula for the following compounds (a) Sodium nitrate (b) Calcium carbonate (c) Lithium hydroxide (d) Potassium carbonate (4) 6. Write the word equations for the following reactions (a) Sodium reacts with water to produce sodium hydroxide and water. (b) Carbon dioxide and water are produced when methane burns completely in oxygen (2) 7. Write a formula equation for the following word equation Copper Carbonate Copper oxide + Carbon dioxide (1) Total = 20 Extension 1. Write the formulae for the following compounds (a) Magnesium Hydroxide (b) Ammonium Phosphate (c) Sodium carbonate Write the ionic formulae for the following compounds (d) Lithium oxide (e) Copper (II) nitrate (5) 2. A strip of rhubarb is found to contain 1.8g of oxalic acid. How many moles of oxalic acid, C2H2O4, are contained in 1.8g (2) 3. Calculate the concentration when 4g of sodium hydroxide dissolves in 250 cm3 of water (2) 4. Urea, H2NCONH2, reacts with water, breaking down to form carbon dioxide and ammonia, NH3.Write the balanced chemical equation for the reaction (2) 5. Rust, iron (III) oxide, that forms on cars can be treated using rust remover which contains phosphoric acid. When painted on, the rust remover changes the iron (III) oxide into iron (III) phosphate. Fe2O3 + H3PO4 FePO4 + H 2O (a) Balance the above equation (1) (b) The rust remover contains 250 cm3 of 2 mol l-1 phosphoric acid, calculate the number of moles of phosphoric acid contained (c) (1) Use your answer from part (b) to calculate the mass of iron (III) oxide that will be removed by 250 cm3 of 2 mol l-1 of phosphoric acid. (3) 6. Silver tarnishes in air forming black silver sulphide, Ag2S. Ag + H2 S + O2 Ag2S + H2 O (a) Balance the above equation (b) Calculate the mass of silver sulphide formed from 1.08g of silver (1) (3) Total = 20