How do you draw it?
advertisement

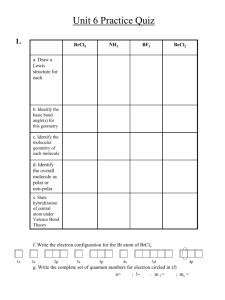
The Shapes of Molecules Or in Big Scientific Words “MOLECULAR GEOMETRY” Simulate Electron Pair Arrangements Use Balloons to represent an Electron Pair What is a pair of electrons going to do to another pair of electrons? THEY REPEL EACH OTHER So if we want to mimic what happens in an atom, we need to get the balloons as far apart as possible. First Some Terminology Nonbonding Pair of Electrons Also called “Lone Pair”, “Unshared Pair” Bonding Pair of Electrons Also called “Shared Pair” 2 Electron Pairs on Central Atom Beryllium fluoride 2 Shared Pairs 0 Unshared Pairs This is a Lewis Structure and a 3D drawing How can we get these electron pairs (balloons) as far apart as possible? What does the shape look like? LINEAR 180º Apart 3 Electron Pairs on Central Atom Boron trihydride 3 Bonding Pairs 0 Nonbonding Pairs This is a 3D drawing of BH3 Get three balloons as far apart from each other. How would you describe the shape? TRIGONAL PLANAR 120º Apart What would it look like if? We had a Double Bond or a Triple Bond? Does a double count as two pairs regardless? Since the two pairs are in the same place do they count as only one? Because they are forced into the same place they only count once. Sulfur dioxide What else new do you notice? 3 Pairs Around Central Atom 2 Shared Pairs Adjusted 1 Lone Pair The lone pair is still there but we can’t see it. Lewis Structure The molecule actually looks like BENT Roughly 120º Apart 3D Shape 4 Pairs Around Central Atom Silicon tetrafluoride Think 3D not 2D… How might you describe shape? TETRAHEDRAL 109.5º Apart Lewis Structure 4 Shared Pairs 0 Lone Pairs How do you draw it? How to Draw a Tetrahedral Shape Regular Lines Mean its in the Plane of the Paper Dashed Line Means its Going Away From You Lewis Structure Bold Line Means its Coming At You. 3D Shape 4 Electron Pairs Around Center What does it look like? Ammonia TRIGONAL PYRAMIDAL Roughly 109.5º Apart Lewis Structure How do you draw it? 3 Bonding Pairs 1 Nonbonding Pair How to Draw Trigonal Pyramidal 4 Electron Pairs around Center What does this one look like? Water BENT Roughly 109.5º Apart How do you draw it? Lewis Structure 2 Shared Pairs 2 Unshared Pairs 3D Shape 5 Electron Pairs! Expanded Octets 5 Shared Pairs Phosphorus pentafluoride What does it look like? 0 Unshared Pairs Lewis Structure TRIGONAL BIPYRAMIDAL Some are 120º Apart. Others are 90º Try to Draw It Six Pairs How do the balloons arrange themselves? Sulfur hexafluoride Lewis Structure OCTAHEDRAL Why “octa”? Octahedron All 90º apart How do you draw it? 6 Shared Pairs 0 Lone Pairs VSEPR Theory Valence Shell Electron Pair Repulsion Electron Pairs try to get as far away from each other as possible.