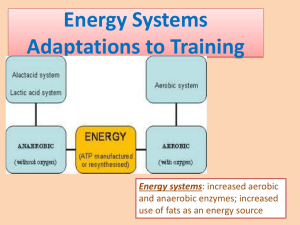
Energy Systems
advertisement

Energy systems Learning outcomes: All are able to demonstrate understanding of the energy sources required for ATP resynthesis All are able to describe the Lactic Acid energy system Most are able to explain the use of PCr in ATP resynthesis Most are able to apply their understanding of the lactic acid system to sporting examples. Some can analyse the lactic acid and determine advantages and disadvantages Starter activity Research has been conducted into “activity cycles” of intermittent sports such as soccer, hockey and rugby, which are reliant on efficient energy systems. a) Identify the principal energy source for each of the following activity cycles in these types of physical activities: (i) walking; (ii) sprinting; (iii) jogging. (3 marks) b) What are the disadvantages of using fat as an energy source during exercise? (2 marks) Answer a) i)Walking – free fatty acid /triglycerides/fats; ii) Sprinting-muscle glycogen/ATP/carbohydrates/PC; ii) Jogging-mixture of fatty acids and muscle glycogen/fats/carbohydrates 3 marks b) 1 Less efficient energy yield per unit of oxygen; 2 Cannot be used anaerobically for sprint type activities/ can only be used aerobically; 3 Requires the presence of carbohydrates to be used; 4 Slow to produce energy/ insoluble in blood. 2 marks Homework List ten different activities where a performer might benefit from taking a creatine supplement. Give reasons in support of your answer Research and make notes on the factors that affect the rate of lactate accumulation: Muscle fibre type Exercise intensity Rate of blood lactate removal Training Respiratory exchange ratio OBLA can be expressed as a percentage of VO2 max. What do you understand by this term and how is it different in trained and untrained performers. What factors affect VO2 max? Energy systems The conversion of these fuels into energy which can then be used to resynthesise ATP occurs through one of 3 energy systems: 1-The ATP-PC System 2- The Lactic Acid System 3- The Aerobic system The Energy Systems Immediate: ATP-PCr Short-term: Lactic acid (glycolysis) Long-term: Aerobic Energy Systems: What we need to know The type of reaction (eg. Aerobic or anaerobic) The chemical or food used (eg. Glycogen) The specific site of the reaction The energy yield (eg. 2ATP from lactic acid system) Specific stages within a system The by-products produced (eg. Lactic acid) The enzyme controlling the reaction. When each system is predominantly used during exercise ATP –PC System ATP-PC system is the first of the anaerobic systems. Energy is released rapidly so used for high intensity maximum work There is a limited store of phosphocreatine in the muscles and can only last for 3 - 10 seconds 100m sprint, performing a vault, smash in tennis, slam dunk ATP-PC System Phosphocreatine (PCr) stored in the sarcoplasm of the muscles The following reaction takes place (facilitated by the enzyme creatine kinase) PCr ------------ Pi + Creatine + Energy This energy is used to recycle ATP Energy + ADP + Pi = ATP These two reactions together are called a coupled reaction Only 1 molecule of ATP can be resythesised by 1 molecule of PC Activity Critically analyse the ATP – PC system. What are the strengths and weaknesses of this system to an athlete. ATP-PC System Advantages Disadvantages ATP resynthesis is very rapid Limited store of PCr in PCr stores recover very quickly (2-3 mins) Anaerobic process No fatiguing by-products Can use creatine supplementation muscle cell, sufficient for 10 secs Fatigue occurs when PCr levels fall and can not sustain ATP resynthesis Resynthesis of PCr needs sufficient oxygen Only 1 molecule of ATP can be resythesised by 1 molecule of PC Quick recap Site of reaction – Fuel used – Active enzyme – Molecules of ATP produced - 100m sprintATP split to drive away from blocks PCr supplies energy for rest of race ATP SPLITTING muscle cell ATP ATPase ATP-PC SYSTEM muscle cell Phosphocreatine Creatine kinase 1 molecule ATP-PC System Tip: Rebuilding or resynthesising ATP from ADP + P is an endothermic reaction (energy is required) Activity Using the pictures demonstrate your understanding of the Lactic Acid system (Anaerobic glycolysis). One person in your group will move to another group to share your understanding and gain further knowledge. Summarise your understanding of the Lactic Acid system. Extension - analyse the system and determine its advantages and disadvantages Lactic Acid System Most activities last for longer than 10 secs. Once phosphocreatine is depleted the lactic acid system (anaerobic glycolysis) takes over and re synthesises ATP from the breakdown of glucose. Glucose is stored in the muscles and liver as glycogen. In order to provide energy to make ATP glycogen has to be converted to glucose. This process is called glycolysis. (Sarcoplasm) Lactic Acid System Glucose is broken down into 6 phosphates (2 ATP) and pyruvic acid. The main enzyme responsible for the break down of glucose is phosphofructokinase (PFK) activated by low levels of phosphocreatine Pyruvic acid is converted into lactic acid in the absence of oxygen. Lactic Acid system Overall summary: C6H12O6 (glucose) 2(C3H6O3) (pyruvic acid) + ENERGY ENERGY 2ADP + 2Pi 2ATP The energy released from the breakdown of each molecule of glucose is used to make two molecules of ATP The lactic acid system actually provides sufficient energy to re-synthesise three molecules of ATP but the process of glycolysis itself requires energy (one molecule) The lactic acid system provides energy for high-intensity activities lasting up to 3 minutes but peaking at 1 minute, for example the 400m Lactic Acid System Advantages Disadvantages Few chemical reactions so If lactic acid accumulates in the ATP can be resynthesises quickly Anaerobic so do not need to wait for the 3 minutes or for sufficient oxygen Lactic acid can be converted back into liver glycogen can be called upon to produce an extra burst of energy (10,000m) muscle, the pH of the body is lowered and this has an effect on enzyme action. PFK, the controlling enzyme, is then inhibited and the ability to regenerate ATP is reduced. This affects performance, for example ‘burning out’ at the end of a race Only a small amount of energy (5%) locked inside a glycogen molecule can be released in absence of oxygen. Quick recap 400m race First 10 secs ATP-PC Lactic Acid will provide for the rest Site of reaction – Fuel used – Active enzyme – Molecules of ATP produced - Sacroplasm of muscle cell Glycogen (stored CHO) Phosphofructokinase 2 molecules Exam Question What are the main energy sources used by an athlete during a 400M sprint? Explain the predominant energy system used during this time. (7 Marks) Make notes on your own to answer this question. Share with the person beside you. Share with the whole class. Candidate A The main energy sources used by a 400m runner are carbohydrate and phosphocreatine. The ATP/PC system is used for the first part of the race and is a simple system to use. It uses phosphocreatine as the fuel and there are no fatiguing by products. The energy yeild is ATP. After 10 seconds the lactic acid system is used. Candidate B The energy sources used by the sprinter are phosphocreatine and glucose. The main energy system is the lactic acid system. This is anaerobic and glucose is broken down into pyruvic acid. Two molecules of ATP are formed and lactic acid is the by product. This system takes place in the sarcoplasm. Energy systems Learning outcomes: All are able to demonstrate understanding of the energy sources required for ATP resynthesis All are able to describe the Lactic Acid energy system Most are able to explain the use of PCr in ATP resynthesis Most are able to apply their understanding of the lactic acid system to sporting examples. Some can analyse the lactic acid and determine advantages and disadvantages Plenary Activity All write down one question and answer that would demonstrate the progress you have made in this lesson.