cancers
advertisement

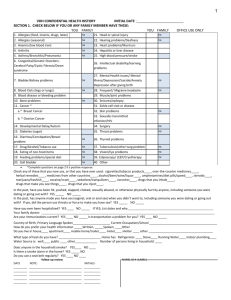
PREVENTEST Final Go Live Training Get Ready to Sell! www.PrevenTests.com Presented by David Gruber National Sales Director, GeneID 914-262-5042 sales@geneidlab.com Conducting Business the “Right” Way GeneID and all of its Distributors and Sales Representatives must comply with all Federal, State, and Local Healthcare Laws, Policies and Procedures This Includes: Anti-Kickback, Stark, and HIIPA GeneID employs a “No Tolerance” approach to compliance violations About GeneID? GeneID is a CLIA and CAP (College of American Pathology) Certified Molecular Laboratory specializing in Next Generation DNA Gene Sequencing using the Ion Torrent Micro Chip Technology by Life Technologies The microchip contains over 140 million mini Test Tubes which allow us to be very accurate, cost effective, do many tests at once and have a very quick turnaround time We are based in Ramsey, NJ and service all states except NY Our staff includes a CEO, Scientific Director, Medical Director, Billing Department, Lab Technicians, Sales Support, Access to Genetic Counselors, and national Field Sales Team About Our Scientific Director Dr. Daniel Cohen, M.D. Double Board Certified M.D. in OB/GYN and Genetics Expert in NGS Human Genome Project BRCA Protocol Lead, Argentina Twice Listed in Who’s Who of Science: 1996 and 2013 Director of 4 molecular laboratories Oversees all testing in the GeneID Laboratory Why Genetic Testing for Cancer Risk is so important… • Patients who have or had cancer caused by a genetic mutation are at significant risk for another related cancer • Family members of patients with cancer are at a higher risk for developing a related cancer • Preventing Cancer is a much better than being treated for cancer Introducing The GeneID PREVENTEST; a New, First-to-Market, non-invasive cancer risk pre-disposition test analyzing 31 Genes linked to 8 of the most common solid tumor hereditary cancers: We are in the Right Space at the Right Time!! 1. The Next Generations Sequencing (NGS) Market is growing exponentially! 2. The Affordable Care Act has made Precision Medicine and Genetic Testing a Priority 3. There is a tremendous investment being made into Research and Technology resulting in more knowledge about which Gene mutations contribute to which cancers and suggesting even more people should be tested 4. Myriad Labs is helping spread the message that “testing more genes in addition to BRCA 1 and 2 results in the detection of more cancers” through their MyRISK test Our Market is Young and Growing!! Expected Market Growth: The next generation sequencing market is rapidly evolving with a large number of developments taking place to increase accuracy and speed, and reduce costs of sequencing. It is the fastest-growing and most lucrative segment in the genomics space with an estimated growth of 23.1%. The global NGS market will be worth $2.5 billion in 2014 and is poised to reach $8.7 billion by 2020. Source: MarketsandMarkets.com (Sept. 2014) http://marketsandmarkets.com/Market-Reports/next-generation-sequencing-ngstechnologies-market-546.html Obama Announces “Precision Medicine Initiative” (Jan. 30, 2015) $217 Million dollar initiative that moves away from one- size-fits-all treatments Scientists will assemble databases of about a million volunteers to study their genetics …to learn how to individualize care. Doctors would study genetics to individualize care Plus…BRCA Testing Granted Preventive Care Designation Under the Affordable Care Act http://tablet.olivesoftware.com/Olive/Tablet/AtlantaJournalConstitution/S haredArticle.aspx?href=AJC/2015/01/31&id=Ar00603 New Clinical Studies Discovering More Gene Mutations that Cause Cancer Published: In the prestigious New England Journal of Medicine August 7th, 2014 (http://www.nejm.org/doi/full/10.1056/NEJMoa1400382) Title: Breast-Cancer Risk in Families with Mutations in PALB2 Results: The absolute breast-cancer risk for PALB2 female mutation carriers by 70 years of age ranged from 33% for those with no family history of breast cancer to 58% for those with two or more first-degree relatives with breast cancer at 50 years of age. ***PALB2 is on our PREVENTEST Panel What is Genetic Testing for Cancer Risk? Predictive testing that can help predict the likelihood that an individual will develop cancer in his or her lifetime Not everyone with a cancer-related gene will develop cancer Who may be at risk for developing hereditary cancer and who should be tested? What criteria determine Medical Necessity? People with Personal history of cancer People with a family member who tested positive for a BRCA mutation People with a Family history of cancer: Two or more relatives on the same side of the family with a related cancer (one must be under age 50) Evolution of Hereditary Cancer Risk Testing… from 2 Genes to MultiGene Panel Testing Early 90’s -BRCA 1 and 2 Discovered by several scientists (Mary Claire King) -Myriad granted a patent on the BRCA 1-2 Genes and has monopoly on market -Myriad launched BRCAnalysis, a predictive medicine product for hereditary breast and ovarian cancer (testing BRCA 1-2) in 1996 -Supreme Court Overturns Myriad BRCA 1-2 Patent in June 2013 opening market to other labs (Ambry, GeneDx, LabCorp, Quest, etc) -Move FROM just testing for BRCA 1&2 to Multiple Gene Testing We are ahead of the Competition… “After the Supreme Court last year ruled that Myriad Genetics could not patent the BRCA1 and BRCA2 genes, the company announced that by 2015 it would replace its BRCAnalysis test with a broader myRisk panel that analyzes 25 genes associated with hereditary cancers, including breast, colorectal, ovarian, endometrial, pancreatic, prostate, gastric, and melanoma” ***And they will be using Next Generation Sequencing! ***Have been piloting for about 6 months ($50m in sales) ***Blood Draw vs. Cheek Swab with PREVENTEST ***25 Genes vs. 31 with PREVENTEST Testing More Genes identifies more Cancers? 2013 San Antonio Breast Cancer Symposium (Myriad Funded Study) It was found that myRisk testing for 25 genes has the ability to identify 51% more patients with higher risk of hereditary breast and ovarian cancer than testing of BRCA1 and BRCA2 genes alone. 2013 Collaborative Group of the Americas on Inherited Colorectal Cancer (CGA) Annual Meeting (Myriad Funded Study) Myriad had reported a 61% improvement in detection of hereditary colon cancer by testing 25 Genes 1. “Why Should I test for Cancer Risk?” 2. “Why would my patients want to know this information?” Because if they know that they are genetically pre-disposed to any of these 8 cancers, they can reduce the risk or even PREVENT the cancer from occurring! What this information allows you to do…Take Control and develop a preventative plan! People at a higher risk for cancer may have the option of having: 1. More frequent cancer screenings 2. Avoiding specific risk factors 3. Making lifestyle changes to lessen additional risk 4. Taking preventive medication (chemoprevention) 5. Having risk reducing surgeries in order to reduce their risk 3. “What do I do with the information?” You do the same thing that you would do if you have an out of control diabetic or blood pressure patient…refer them to a Specialist…a Genetic Counselor 1. First bring patient back into office and review results 2. If positive refer them to a local Genetic Counselor or utilize Telephone Consultation Genetic Counselor experts (Informed DNA) The Panel of Genes What Myriad is Missing Gene ELAC2 HRAS1 MRE11A PTCH1 RAD50 RET Breast Ovarian Colorectal Endometrial Melanoma Pancreatic Gastric Prostate Other ● ● ● ● ● ● ● ● ● • Meta-analysis of associations of the ser217-to-leu and ala541-to-thr variants in ELAC2 (HPC2) and prostate cancer. Camp NJ et al. Am. J. Hum. Genet. 71: 1475-1478, 2002. • Association of HPC2/ELAC2 genotypes and prostate cancer. Rebbeck TR et al. Am. J. Hum. Genet. 67: 1014-1019, 2000. • Cancer Epidemiol Biomarkers Prev. 2003 Dec;12(12):1528-30.The HRAS1 variable number of tandem repeats and risk of breast cancer.Tamimi RM, Hankinson SE, Ding S, Gagalang V, Larson GP, Spiegelman D, Colditz GA, Krontiris TG, Hunter DJ • Damiola, F., et al., Rare key functional domain missense substitutions in MRE11A, RAD50, and NBN contribute to breast cancer susceptibility: results from a Breast Cancer Family Registry case-control mutation-screening study. Breast Cancer Res, 2014. 16(3): p. R58 • http://ghr.nlm.nih.gov/gene/PTCH1 • Screening for BRCA1, BRCA2, CHEK2, PALB2, BRIP1, RAD50, and CDH1 mutations in high-risk Finnish BRCA1/2-founder mutation-negative breast and/or ovarian cancer individuals. Kuusisto KM et al. Breast Cancer Research 2011, 13:R20. • Hum Genet. 2005 Jul;117(2-3):143-53. A newly identified RET proto-oncogene polymorphism is found in a high number of endocrine tumor patients. Gartner W, Mineva I, Daneva T, Baumgartner-Parzer S, Niederle B, Vierhapper H, Weissel M, Wagner L. One Mutation can increase the likelihood of many cancers Performance Goals Ramp up to 50-100 Paid Tests/Month minimum May: 10-15 Tests June: 25-50 Tests And Upward from There! Where Does Your Business Come From? 1. PCPs (40+ tests/month) 2. OB/GYNs (20+ tests/Doc/Month) 3. General Oncologists (40+ tests/month) 4. Breast Surgeons (40+ tests+/month) 5. Others: Large Practices (GI, Wellness, etc.) Good Screening is the Key to Success! Related Cancers (2 categories) BRCA Cancers Lynch Cancers •Breast •Ovarian •Prostate •Pancreas • Colon/Colorectal • Uterus/Endometrial • Pancreas • Ovarian • Stomach/Gastric • Brain • Kidney Qualified Patient Criteria Combinations Female Personal History of: Breast or Ovarian cancer (any age) Colon Cancer (< 60) Female with Family History of: 1 Relative w/Ovarian cancer (any age) 1 Relative w/Breast cancer (diagnosed <45) 2 or more Relatives with Breast, Prostate and/or Pancreatic cancer with 1 diagnosed <50 2 or more Relatives with Colon, Uterus, Pancreatic, Stomach, Brain and/or Kidney cancer with 1 diagnosed <50 3 or more Relatives with any of the above groups of cancer diagnosed at any age Relatives need to be on same side of family Female Other: Patient is Ashkenazi Jewish with 1 Relative w/Breast cancer (any age) Male must have Personal History of Cancer Male Personal History of: Colon Cancer (< 60) Colon (>60), Pancreas, Stomach, Brain and/or Kidney cancer PLUS 1 family member with a related cancer diagnosed at any age Prostate Cancer Plus 2 family members with any combination of Prostate, Breast, Ovarian, and/or Pancreatic cancer (any age) Male Breast Cancer Cancer History that does NOT Qualify 1. Lung Cancer 2. Skin Cancer 3. Liver Cancer 4. Bone Cancer 5. Throat Cancer 6. Esophageal Cancer 7. Heart Cancer 8. Cervical Cancers 9. Thyroid 10. Blood Cancers (Leukemia, Lymphoma, Hodgkin's) Some key rules to Remember • For Medicare, Males with NO PERSONAL History does not qualify • Colon and Breast are not “Related Cancers” • Skin is not “In” and does not count as a qualifier • Family Members need to be on same side of family and 1st or 2nd degree relatives (mother, father, sister, brother, aunt, uncle, niece, nephew, ½ brother/sister) Requisition Form Very Important: Fill in All Applicable Information -Need Ages of Cancer Diagnosis -Need Doc Sig -Need Patient Sig S -Need Insurance Info Write your name on the top of the Req form ICD 9 Codes for Personal History “Does This Test Qualify?” “Does This Test Qualify?” “Does This Test Qualify?” “Does This Test Qualify?” The “Kit” New Non-Invasive Sample Collection Method To Complete Kit: Pick up softpack FedEx Express envelopes and insert in kits and leave with offices Marketing/Support Materials -PREVENTEST Website www.PrevenTests.com -Video illustrating “PREVENTEST” (Great to mail to potential customers) PREVENTEST 2 Minute Video Description in English: https://vimeo.com/96718022 PREVENTEST 2 Minute Video Description in Spanish: https://vimeo.com/112819235 -Sales Support -Dropbox with sales materials -Sales Status Reports once/week -Test Kits and Patient Education leave behinds -Genetic Counseling Referral company The “New Client” Form Fill out completely and send to you distributor lead: Or fax to lab: (201) 825-0191 This is what links the Doctor/Office to you so you will get credit for the tests that are sent into the lab Keys to Success Keep it Simple with your customers…so don’t overcomplicate the message by trying to talk “high science” Our message is: “Doctor, with NGS technology, you can now, with a simple cheek swab, predict the risk of 8 serious solid tumor cancers so that your patients can put a preventative plan in place” Don’t mistake “Excitement” for “Commitment” High Frequency on a few (3-5) Offices until they routinely send tests #1 Key to Success Make Screening for Cancer a “standard” in the office (just like they screen for cholesterol and blood pressure) USE THE CUSTOMIZABLE OFFICE POSTERS and PATIENT QUESTIONNAIRE to profile the most appropriate candidates Office Posters to be placed in waiting room and exam rooms to generate awareness) (Version 1) Office Posters to be placed in waiting room and exam rooms to generate awareness (Version 2) (Version 2) Questionnaire to determine if patient qualifies (version 1) To be included on clipboard for all patients coming in for appointment Can be emailed or mailed to patients in practice database Questionnaire to determine if patient qualifies (version 2) To be included on clipboard for all patients coming in for appointment Can be emailed or mailed to patients in practice database Feedback Mechanism/Other Suggestions Ask Office to text or call you when a test is coming in and let your distributor know Conference Calls to share successes (GeneID call every Friday at 11:15AM EST) Encourage Multiple tests/Fed Ex shipment Request additional kits through your distributor (New Kits will only be sent once we receive 5 tests) To the Dropbox… Selling Materials