Ppt
advertisement

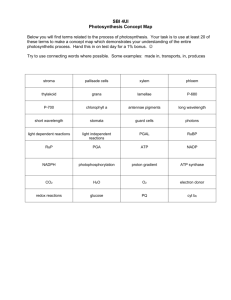
Chapter 4: Energy From the sun to you in just two steps Lectures by Mark Manteuffel, St. Louis Community College Learning Objectives Understand and be able to explain the following: How energy flows from the sun and through all life on earth How photosynthesis uses energy from sunlight to make food Learning Objectives Understand and be able to explain the following: How cellular respiration converts food molecules into ATP, a universal source of energy Alternative pathways to energy acquisition Energy flows from the sun and through all life on earth. 4.1 Cars that run on french fry oil? Organisms and machines need energy to work. What are biofuels? Q Humans can get energy from food. Can machines? Biofuels and Fossil Fuels Chains of carbon and hydrogen atoms • Energy is stored in the bonds Animal fats and oils How do fuels provide energy? The activities of living organisms are fueled by breaking chemical bonds and harnessing the released energy. Energy Conversions All life depends on capturing energy from the sun and converting it into a form that living organisms can use. Two key processes • Photosynthesis • Cellular respiration Take-home message 4.1 The sun is the source of the energy that powers all living organisms and other “machines.” The energy from sunlight is stored in the chemical bonds of molecules. Take-home message 4.1 When these bonds are broken, energy is released, regardless of whether the bond is in a molecule of food, of a fossil fuel or of a biofuel such as the oil in which french fries are cooked. 4.2 Energy has two forms. Kinetic and Potential What is energy? The capacity to do work Work • Moving matter against an opposing force Kinetic Energy The energy of moving objects • Heat energy • Light energy Potential Energy A capacity to do work that results from the location or position of an object Concentration energy Food gradients and potential has potential energy Chemical Energy Take-home message 4.2 Energy, the capacity to do work, comes in two forms. Take-home message 4.2 Kinetic energy is the energy of moving objects. Potential energy, such as chemical energy, is stored energy that results from the position or location of an object. 4.3 As energy is captured and converted, the amount of energy available to do work decreases. Energy Conversions Only ~1% of the energy released by the sun that earth receives is captured and converted by plants. • Converted into chemical bond energy What happens to the other 99%? Thermodynamics The study of the transformation of energy from one type to another First Law of Thermodynamics Energy It can never be created or destroyed. can only change from one form to another. Energy Tax! Every time energy is converted from one form to another the conversion isn’t perfectly efficient. Some of the energy is always converted to the least usable form of kinetic energy: heat. Second Law of Thermodynamics Every conversion of energy includes the transformation of some energy into heat. Heat is almost completely useless to living organisms Take-home message 4.3 Energy is neither created nor destroyed but can change form. Each conversion of energy is inefficient, and some of the usable energy is converted to less useful heat energy. 4.4 ATP molecules are like free-floating rechargeable batteries in all living cells. How do cells directly fuel their chemical reactions? None of the light energy from the sun can be used directly to fuel cellular work. First it must be captured in the bonds of a molecule called adenosine triphosphate (ATP). Structure of ATP ATP Molecules Cells cannot use light energy directly to do work. First, the energy has to be converted into chemical energy in ATP molecules. Adenosine Triphosphate Pop off the third phosphate group • ATP ADP + Phosphate group + energy release Release a little burst of energy! Use this energy to drive chemical reactions necessary for cellular functioning. • Building muscle tissue • Repairing a wound • Growing roots Recycling in the Cell ADP + phosphate group + energy = ATP Take-home message 4.4 Cells temporarily store energy in the bonds of ATP molecules. This potential energy can be converted to kinetic energy and used to fuel life-sustaining chemical reactions. At other times, inputs of kinetic energy are converted to the potential energy of the energyrich but unstable bonds in the ATP molecule. Photosynthesis uses energy from sunlight to make food. 4.5 Where does plant matter come from? Photosynthesis: the big picture. From a seed to a tree: Where does the mass come from? Photosynthetic Organisms Photosynthesis: The Big Picture 3 inputs 2 products Take-home message 4.5 Through photosynthesis, plants use water, the energy of sunlight, and carbon dioxide gas from the air to produce sugars and other organic materials. In the process, photosynthesizing organisms also produce oxygen, which makes all animal life possible. 4.6 Photosynthesis take place in the chloroplasts Organelles found in plant cells A Closer Look at Chloroplasts Take-home message 4.6 In plants, photosynthesis occurs in chloroplasts, green organelles packed in cells near the plants’ surfaces, especially in the leaves. 4.7 Light energy travels in waves: plant pigments absorb specific wavelengths Light Energy A type of kinetic energy Made up of little energy packets called photons Light Energy Different photons carry different amounts of energy, carried as waves. Length of the wave = amount of energy the photon contains. Electromagnetic Spectrum Range of energy that is organized into waves of different lengths. Shorter the wavelength, higher the energy. Visible Spectrum Range of energy humans see as light ROYGBIV Pigments = molecules that absorb light Chlorophyll Plant pigment Absorbs certain wavelengths of energy (photons) from the sun Absorbed energy excites electrons Plant Pigments Plant pigments can only absorb specific wavelengths of energy Therefore, plants produce several different types of pigments Plant Pigments Chlorophyll a Chlorophyll b Carotenoids Take-home message 4.7 Photosynthesis is powered by light energy, a type of kinetic energy made up from energy packets called photons. Take-home message 4.7 Photons hit chlorophyll and other lightabsorbing molecules in cells near the green surfaces of plants. These molecules capture some of the light energy and harness it to build sugar from carbon dioxide and water. 4.8 Photons cause electrons in chlorophyll to enter an excited state. Electron Excitation Conversion of electromagnetic energy into chemical energy of bonds between atoms Photons of specific wavelengths bump electrons up a quantum level into an excited state Two Potential Fates of Excited Electrons (1) Electrons return to their resting, unexcited state. (2) Excited electrons are passed to other atoms. The Passing of Electrons in Their Excited State Chief way energy moves through cells Molecules that gain electrons always carry greater energy than before receiving them • Can view this as passing of potential energy from molecule to molecule Take-home message 4.8 When chlorophyll gets hit by photons, the light energy excites an electron in the chlorophyll molecule, increasing the chlorophyll’s potential energy. The excited electrons can be passed to other atoms, moving the potential energy through the cell. 4.9 Photosynthesis in detail: the energy of sunlight is captured as chemical energy. FOLLOW THE ELECTRONS! The “Photo” Part Sunlight ATP A high-energy electron carrier Electrons That Leave the Photosystem Are Replenished Where does oxygen come from? An Electron Transport Chain Connects the two photosystems Product #1 of the “Photo” Portion of Photosynthesis: ATP The Second Photosystem Follow the electrons Product #2 of the “Photo” Portion of Photosynthesis: NADPH Products from the “Photo” Portion ATP and NADPH Time for the “synthesis” part! Take-home message 4.9 There The are two parts of photosynthesis. first is the “photo” part, in which light energy is transformed into chemical energy, while splitting water molecules and producing oxygen. Take-home message 4.9 Sunlight’s energy is first captured when an electron in chlorophyll is excited. As this electron is passed from one molecule to another, energy is released at each transfer, some of which is used to build the energy-storage molecules ATP and NADPH. “SYNTHESIS” 4.10 Photosynthesis in detail: the captured energy of sunlight is used to make food. The Calvin Cycle Series of chemical reactions Occurs in stroma Enzymes are recycled The Processes in the Calvin Cycle Occur in Three Steps: Take-home message 4.10 The second part, or “synthesis” part, of photosynthesis is the Calvin cycle, and it occurs in the stroma of the chloroplast. Take-home message 4.10 During this phase, carbon from CO2 in the atmosphere is attached (fixed) to molecules in chloroplasts, sugars are built, and molecules are regenerated to be used again in the Calvin cycle. The fixation, building, and regeneration processes consume energy from ATP and NADPH (the products of the “photo” part of photosynthesis). 4.11 The battle against world hunger can use plants adapted to water scarcity. Evolutionary Adaptations Some plants thrive in hot, dry conditions Adaptations that reduce evaporative water loss • How do plants use water? Stomata Pores for gas exchange How to get CO2 when stomata are shut? C4 Photosynthesis • C4 plants produce ultimate “CO2-sticky tape” enzyme. • C4 photosynthesis adds an extra set of steps. CAM Photosynthesis Close stomata during hot dry days At night, stomata open, CO2 let in and temporarily bound to a holding molecule During day, CO2 gradually released and used while stomata are closed All Three Photosynthetic Pathways Take-home message 4.11 C4 and CAM photosynthesis are evolutionary adaptations at the biochemical level that, although more energetically expensive than regular (C3) photosynthesis, allow plants in hot, dry climate to close their stomata and conserve water without shutting down photosynthesis. Cellular respiration converts food molecules into ATP, a universal source of energy for living organisms. 4.12 How do living organisms fuel their actions? Cellular respiration: the big picture. Cellular Respiration The big picture A Human Example Eat food Digest it Absorb nutrient molecules into bloodstream Deliver nutrient molecules to the cells At this point, our cells can begin to extract some of the energy • stored in the bonds of the food molecules Take-home message 4.12 Living organisms extract energy through a process called cellular respiration, in which the high-energy bonds of sugar and other energy-rich molecules are broken, releasing the energy that went into creating them. Take-home message 4.12 The cell captures the food molecules’ stored energy in the bonds of ATP molecules. This process requires fuel molecules and oxygen, and it yields ATP molecules, water, and carbon dioxide. 4.13 The first step of cellular respiration: glycolysis is the universal energy-releasing pathway. Glycolysis: the universal energy-releasing pathway Glycolysis Three of the ten steps yield energy – quickly harnessed to make ATP. High-energy electrons are transferred to NADH. Net result: each glucose molecule broken down into two molecules of pyruvate ATP molecules produced NADH molecules store high-energy electrons Take-home message 4.13 Glycolysis is the initial phase in the process by which all living organisms harness energy from food molecules. Glycolysis occurs in a cell’s cytoplasm and uses the energy released from breaking chemical bonds in food molecules to produce high-energy molecules, ATP and NADH. 4.14 The second step of cellular respiration: the Krebs cycle extracts energy from sugar. The Preparatory Phase to the Krebs Cycle Payoff from the Krebs cycle: ATP NADH FADH2 Take-home message 4.14 A huge amount of additional energy can be harvested by cells after glycolysis. First the end-product of glycolysis, pyruvate, is chemically modified. Then, in the Krebs cycle, the modified pyruvate is broken down step-by-step. Take-home message 4.14 This breakdown process releases carbon into the atmosphere (as CO2) as bonds are broken, and captures some of the released energy in two ATP molecules and numerous high-energy electron carriers for every glucose molecule. 4.15 The third step in cellular respiration: ATP is built in the electron transport chain. Mitochondria Two key features of mitochondria are essential to their ability to harness energy from molecules: • Feature 1: mitochondrial “bag-within-a-bag” structure • Feature 2: electron carriers organized within the inner “bag” The “bag-withina-bag” Follow the Electrons… (just as we did in photosynthesis) #2) This proton concentration gradient represents a significant source of potential energy! Proton Gradients and Potential Energy The force of the flow of H+ ions fuels the attachment of free-floating phosphate groups to ADP to produce ATP. Take-home message 4.15 The largest energy payoff of cellular respiration comes as electrons from NADH and FADH2 produced during glycolysis and the Krebs cycle move along the electron transport chain. Take-home message 4.15 The electrons are passed from one carrier to another and energy is released, pumping protons into the intermembrane space. As the protons rush back to the inner mitochondrial matrix, the force of their flow fuels the production of large amounts of ATP. 4.16 THIS IS HOW WE DO IT Can we combat the fatigue and reduced cognitive functioning of jet lag with NADH pills? Pure or Basic Science and Applied Science • What is jet lag and why is it of scientific interest? • How is jet lag related to cellular respiration? Why should NADH alleviate symptoms of jet lag? • If levels of NADH could be increased by taking the molecule in pill form, this might lead to increased production of usable energy through the electron transport chain. • Testable prediction: – “Supplementing NADH should counteract some of the effects of jet lag, including reduced cognitive functioning and fatigue.” Experimental Setup • The researchers used a randomized, controlled, double-blind experimental design. • 2 groups: Placebo or NADH • Battery of tests • Overnight “red-eye” flight • Retesting Did NADH reduce the symptoms of jet lag? 1. Vigilance Placebo: 37% of subjects made omission errors. NADH: 14% of subjects made omission errors. Did NADH reduce the symptoms of jet lag? 2. Working memory Placebo: Subjects answered 6.8 more problems per minute than in the baseline test. NADH: Subjects answered 13.2 more problems per minute than in the baseline test. Did NADH reduce the symptoms of jet lag? 3. Multi-tasking Placebo: Subjects increased performance by 19.2 points over baseline. Subjects’ reaction time was slower than baseline by 0.44 seconds. NADH: Subjects increased performance by 77.5 points over baseline. Subjects’ reaction time was faster than baseline by 0.15 seconds. Did NADH reduce the symptoms of jet lag? 4. Visual perception Placebo: Subjects completed 1.4 more items per minute than at baseline. NADH: Subjects completed 5.4 more items per minute than at baseline. Did NADH reduce the symptoms of jet lag? 5. Sleepiness Placebo: 75% of subjects reported increased sleepiness. NADH: 25% of subjects reported increased sleepiness What conclusions can we draw from these results? • The researchers’ conclusion, supported by the evidence, was that “NADH appears to be a suitable short-term countermeasure for the effects of jet lag on cognition and sleepiness.” Take-home message 4.16 • The symptoms of jet lag—including fatigue, memory loss, and reductions in cognitive performance—can impair the performance of people in many professions today. Take-home message 4.16 • The results of a randomized, controlled, double-blind study support the hypothesis that an NADH supplement may be a suitable short-term countermeasure for these effects. There are alternative pathways to energy acquisition. 4.17 Beer, wine, and spirits are byproducts of cellular metabolism in the absence of oxygen. Take-home message 4.17 Oxygen deficiency limits the breakdown of fuel because the electron transport chain requires oxygen as the final acceptor of the electrons generated during glycolysis and the Krebs cycle. Take-home message 4.17 When oxygen is unavailable, yeast resort to fermentation, in which they use a different electron acceptor, pyruvate, and in the process generate ethanol, the alcohol in beer, wine, and spirits. 4.18 Eating a complete diet: cells can run on protein and fat as well as on glucose. Take-home message 4.18 Humans and other organisms have metabolic machinery that allows them to extract energy and other valuable chemicals from proteins, fats, and carbohydrates in addition to the simple sugar, glucose. Learning Objectives Understand and be able to explain the following: How energy flows from the sun and through all life on earth How photosynthesis uses energy from sunlight to make food How cellular respiration converts food molecules into ATP, a universal source of energy Alternative pathways to energy acquisition