AP Biology Essential Chemistry
advertisement

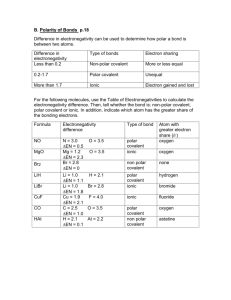
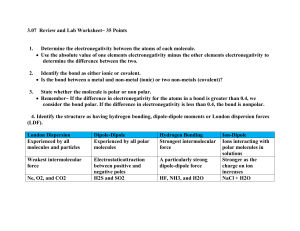
To determine the types of bonds, you have to find out the atom’s electronegativity. (attraction for electrons) Fewer electron shells= greater electron negativity More electron shells= less electron negativity Electronegativity Trends To determine whether bonds are ionic, polar covalent, or non-polar covalent, you need to figure out the difference in electronegativity between the atoms that make up a molecule -If the difference between 2 atom’s electronegativity is small, the bond is likely to be non-polar covalent (difference is less than 0.5) -If the difference between 2 atom’s electronegativity is larger, the bond is more likely to be ionic (difference is greater than 1.6) -If the values are about the same, they produce polar covalent bonds (difference is between 0.5 and 1.6) NH3 is extremely soluble in the aqueous solution in a cell O2 is relatively insoluble Ammonia is polar covalent with its bond because the electronegativity of N is 3.0 and H is 2.1. When subtracted their total is .9. Since the water molecule is also polar, the ammonia will dissolve easily. The hydrogen in the ammonia will unbond from the nitrogen and rebond to the water molecules. http://www.johnkyrk.com/cellmembrane.ht ml Ammonia Fountain Experiment Oxygen gas is non-polar covalent because the two oxygen atoms have the same electronegativity, so when subtracted they equal zero. Since water is polar and oxygen gas is not, it will not dissolve easily. Oxygen gas consists of two oxygen atoms and when bonded together they have eight electrons in their valence shells, making them “happy.” Therefore they have no interest in rebonding with the water molecules. Here’s just a fun video about oxygen and water.