Molecular Shapes - Del Mar College
advertisement

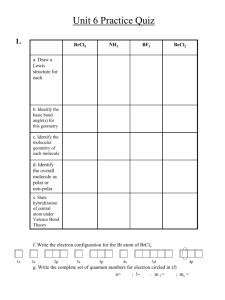
Molecular Shapes Lewis Diagram Allow You to Determine the Three-dimensional Shape of a Molecule Molecular Shape is Important! Shape influences physical properties – Affects features such as boiling point, physical state, hardness, etc – Water, for example, would surely be a gas at room temperature (and well below) if its shape were linear – Instead water’s bent shape makes it a liquid at ordinary temperatures Water H O H If water had a linear shape, it would be a gas! Water H H O H If water had a linear shape, it would be a gas! O H Bent water is, of course, a liquid; shape matters! Molecular Shape is Important Shape influences chemical properties – Affects rate of reaction – Affects site within a molecule where reaction might occur – Affects type of catalyst or enzyme which might be operative with reaction Determining a Molecule’s Shape: It’s as Easy as 1-2-3 Actually, not quite... It’s as easy as 2-3-4, instead! We only need to count GROUPS of electrons around certain atoms to determine shape in the vicinity of that atom Electron Groups An electron group is a set of electrons that are in the same area of the molecule – – – – The two electrons of a lone pair The two electrons of a single bond The four electrons of a double bond The six electrons of a triple bond Each of the above counts as a single electron group Electron Groups Electron Groups Electron Groups Electron Groups Electron Groups Four groups of Electrons Electron Groups H H N: H Electron Groups H one group H N: H Electron Groups one group H one group H N: H Electron Groups one group H one group H N: one group H Electron Groups one group H one group H one group N: one group H Four groups of electrons. Lone pairs count! Electron Groups H .. O: H Electron Groups one group H .. O: H Electron Groups one group H one group .. O: H Electron Groups one group H one group one group .. O: H one group Again, lone pairs count. Electron Groups There are three electron groups around carbon. Double bond counts as one group. Electron Groups one group H C N: one group Around carbon, there are only two electron groups; the triple bond is one group. Determining Shapes Use the Lewis diagram to establish shape The shape is described at each central (pivot, “hinge”) atom in the molecule Some molecules may have different shapes at different central atoms Don’t worry about shape at outside atoms Our molecules will usually have only one central atom Determining Shapes H .. O: H outside atom (no shape) central atom (shape important) Determining Shapes . . CN H shape not important (outside atoms) shape important (central, pivot atoms) Determining Shapes The number of electron groups around a central atom determines its shape The shape will correspond to a 3-D arrangement which allows maximum separation of each electron group Remember, an electron group is: – a lone pair – a bond group (single, double, triple bond) Two electron groups For octet-rule atoms (ours are!), this will be either – one single bond + one triple bond HCN: 2 electron groups Two electron groups For octet-rule atoms (ours are!), this will be either – one single bond + one triple bond – two double bonds HCN: 2 electron groups .. .. :O=C=O: 2 electron groups Two electron groups Two electron groups generate a LINEAR (straight-line) geometry (180°) separation Atoms connected to the linear electron groups lie in their same direction .. .. :O=C=O: HCN: 180° separation 180° separation = linear Three Electron Groups For octet-rule atoms this combination will be: – two single bonds + a double bond – one single bond + one double bond + one lone pair one group one group one group (lone pairs at O, Cl omitted, for clarity) Three Electron Groups Three electron groups generate a TRIGONAL (triangle-like) geometry – electron groups and their atoms are about 120° apart Four Electron Groups Result – – – – from: four single bonds three single bonds + a lone pair two single bonds and two lone pairs never multiple bonds Four Electron Groups Result from: – four single bonds Four Electron Groups Result from: – four single bonds – three single bonds + a lone pair 4 groups, including lone pair Four Electron Groups Result from: – four single bonds – three single bonds + a lone pair – two single bonds and two lone pairs .. H 4 groups: 4 groups, including lone pair count ‘em... Four Electron Groups 4 Electron groups generate a TETRAHEDRAL geometry, named after tetrahedron, a figure from 3-D geometry 109° X It is as if the tetrahedral atom were “buried” in the tetrahedron. An angle of about 109° exists between corners and the central atom Four groups of Electrons Four groups of Electrons N Four groups of Electrons O H H