Chapter 1 Chemistry Test Review
advertisement

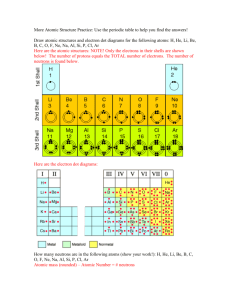
Chemistry Part 2 Test Review for Friday the 13th! The Atom: Label the parts proton, neutron, electron, nucleus, electron cloud on the atom diagram below. - + + + + + Bohr’s Model: Maximum number of electrons in each energy level: ______ 1st level ______ 2nd level ______ 3rd level ______ 4th and higher Which element is this Bohr model representing? _________________ Can you have a level not be “full” before going to the next? _________________ How many valence electrons does the element have? _________________________________________ Is it stable or unstable? ______________ What does it mean for an element to be stable? _____________________________________________________________________________ _____________________________________________________________________________ What group and period is this element in? _________________________________________ Using Periodic Table: What is the atomic number of cobalt? _________ What is the atomic mass of iodine? ________ What are the columns called in the Periodic Table? _____________________ Where are most of the nonmetal elements?(circle one) Left of zigzag Where are most of the metal elements?(circle one) Left of zigzag Right of zigzag Right of zigzag On zigzag On zigzag Using Periodic Table Continued: What is the symbol for molybdenum? __________ In what group can you find strontium? _________ What do the elements Barium, Gold, Platinum, and Mercury have in common? ____________________ ____________________________________________________________________________________ What is the name of the scientist credited for developing the first periodic table? ___________________ ____________________________________________________________________________________ Determining Protons, Neutrons, and Electrons: Which three numbers that deal with atoms are always the same number? (circle 3 below) Atomic number Atomic mass # of protons # of neutrons # of electrons What two numbers do you add together to get the atomic mass? (circle 2 below) Atomic number Atomic mass # of protons # of neutrons # of electrons Which two numbers do you subtract to find the number of neutrons? (circle 2 below) Atomic number Atomic mass # of protons How many protons does mercury have? _______ How many neutrons does zirconium have? _______ How many electrons does tin have? _______ # of neutrons # of electrons How many neutrons does radon have? _______ How many electrons does lanthanum have? _______ How many protons does platinum have? _______ Electron (Lewis) Dot Diagrams: Write the electron dot diagrams for the elements below. Strontium Selenium Xenon Potassium What do the dots represent around each of the symbols in the Lewis Dot Diagram? __________________________________________________________________ Draw Bohr Models for the following Elements: Lithium: Boron: Silicon: