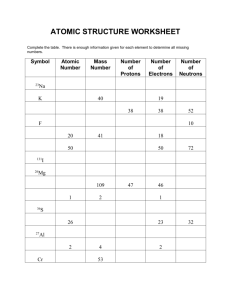
4.1 – Average Atomic Mass
advertisement

4.1 – Average Atomic Mass Average Atomic Mass – the _________ _________ of naturally occurring isotopes of an element (this is what is found on the periodic table) **isotopes of elements are found in varying amounts, which is why the atomic mass you find on the periodic table is always a decimal. The mass on the periodic table is not the mass of a single atom, but instead the average of all atoms of that element. Copper has three naturally occurring isotopes. Copper-63 makes up 69.15% of copper, weighing 62.93 amu. Copper-65 makes up 30.85% of copper at 64.93 amu. What is the average atomic mass of the isotopes? Silver is composed of two naturally occurring isotopes, Silver-107 and Silver-109. Silver-107 has a mass of 106.90 amu and exists at 51.86% of the element. Silver-109 has a mass of 108.90 amu and comprises the remaining amount of Silver. What is the average atomic mass? Silcon has three naturally occurring isotopes. Silcon-28 has a mass of 27.98 amu, Silicon-29 has a mass of 28.98 amu, and Silicon-30 has a mass of 29.97 amu. If Silicon-28 makes up 92.21%, Silicon-29 makes up 4.70%, and Silicon-30 makes up 3.09%, what is the average atomic mass? Practice 1. What is average atomic mass of Lithium if 7.42% exists as 6Li (6.015 amu) and 92.58% exists as 7Li (7.016 amu)? 2. Magnesium has three naturally occuring isotopes. 78.70% of Magnesium atoms exist as Magnesium-24 (23.9850 amu), 10.03% exist as Magnesium-25 (24.9858 amu) and 11.17% exist as Magnesium-26 (25.9826 amu). What is the average atomic mass of Magnesium? 3. Neon has two major isotopes, Neon-20 and Neon-22. Out of every 250 neon atoms, 225 will be Neon-20 (19.992 amu), and 25 will be Neon-22 (21.991 amu). What is the average atomic mass of Neon? 4. What is the atomic mass of Hafnium if out of every 200 atoms, 10 have mass 176.00 amu, 38 have mass 177.00 amu, 54 have mass 178.00 amu, 28 have mass 179.00 amu, and 70 have mass 180.00 amu? 5. In a sample of 200 Chlorine atoms, it is found that 151 are 35Cl (34.969 amu), and 49 are 37Cl (36.966 amu). What is the average atomic mass of Chlorine? 6. Without doing any math, are there more Bromine-79 atoms or more Bromine-80 atoms on earth? (Hint: look at the periodic table.)