Titration Lab Day
advertisement

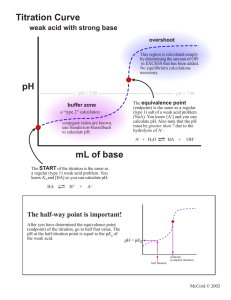
Friday, May 9th: “A” Day Monday, May 12th: “B” Day Agenda Collect Virtual /Titration Practice worksheets Check lab write-ups Lab Discussion Lab: “Titration of an Acid and a Base” Lab will be due next time… Don’t forget extra credit opportunity… Next time… Chapter 15 Review Lab: “Titration of an Acid and a Base” 1. Which acid and base will you be using? HCl and NaOH 2. Are these strong or weak? Strong Lab: “Titration of an Acid and a Base” 3. Which is the titrant? HCl A titrant is a solution of known concentration that is used to titrate a solution of unknown concentration. Lab: “Titration of an Acid and a Base” 4. What indicator will you be using? Phenolphthalein 5. What color is it in a basic solution? Pink Lab Hints Practice controlling the stopper with just water – you need enough control so that the buret can deliver 1 drop. You DO NOT have to refill the burets each time. Simply record the starting and ending volume each time to get the total amount of acid/base added. If you overshoot the end point, don’t worry. Simply add more acid or base and re-titrate until only 1 drop changes the color. Safety Guidelines Both acids and bases are corrosive – safety goggles, aprons, and appropriate shoes must be worn. Be sure to lower the burets to eye level before filling them. Lab: “Titration of an Acid and a Base” Calculation #4: Absolute error: Ea = average Normality(B) – actual value Calculation #5: Percentage error: Er = Ea X 100% actual value Lab: “Titration of an Acid and a Base” You should have time to do 3 trials and complete the calculations today. Be sure to update your table of contents and don’t forget your reflection statement… Be safe and have… Extra Credit Opportunity Friday, 7:00 pm Saturday, 7:00 pm Sunday, 2:00 pm Have me sign your program 15 extra credit points!