Review with answers
advertisement

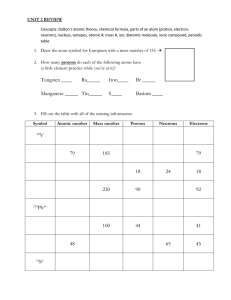
Chapter 4 Study Guide For each of the statements below, write True or False. ___False___ 1. The number of neutrons in an atom is referred to as its atomic number ____True__ 2. The periodic table is arranged by increasing atomic number ___True___ 3. The atomic number is equal to the number of electrons in an atom ___True___ 4. The number of protons in an atom identifies it as an atom of a particular element ___False___ 5. Most atoms have either a positive or a negative charge Answer the following questions. 6. Lead has an atomic number of 82. How many protons and electrons does lead have? 82 Protons and 82 Electrons 7. Oxygen has 8 electrons. How many protons does oxygen have? 8 Protons 8. Zinc has 30 protons. What is its atomic number? 30 9. Astatine has 85 protons. What is its atomic number? 85 10. Rutherfordium has an atomic number of 104. How many protons and electrons does it have? 104 protons, 104 electrons 11. Polonium has an atomic number of 84. How many protons and electrons does it have? 84 protons, 84 electrons 12. Nobelium has an atomic number of 102. How many protons and electrons does it have? 102 protons, 102 electrons Determine the number of protons, electrons, and neutrons for each isotope described below. 13. An isotope has atomic number 19 and mass number of 39 P-19, E-19, N-20 14. An isotope has 14 electrons and a mass number of 28 p-14, e-14, n-14 15. An isotope has 21 neutrons and a mass number of 40 p-19, e-19, n-21 A World-Famous Table Atomic number 1 H Hydrogen 1 Au ______1_____ Atomic mass Gold _____1______ _ 197 Element Name 12 79 Atomic Mass 28 Mg __Magnesium Ni Magnesium Atomic number Nickel 24 Fe 59 ______12___ # protons 26 11 Na _______26___ Iron Element Symbol Sodium 56 _______Fe_____ 23 Atomic number 5 B Boron 11 _____5____ Element Name ______boron # Electrons _____79_______ # Protons _____79_______ # Atomic Number _______79_____ Element Name ___Gold_____ 78 Pt Platinum 195 Pb Atomic number 29 _______117__ ______82____ __ # Electrons 91 88 # Neutrons 207 Zirconium Element Symbol ______Na_____ # Neutrons _____12______ Element Name ___Sodium_ _____78_____ # protons Zr ______Ni___ ____Lead__ Lead 40 __59________ Symbol Element name 82 ___40_______ Atomic Mass ________91__ Atomic number Ra ______88____ Radon # Neutrons 226 _____138__ Atomic mass Cu Copper 64 _____64____ # Neutrons ______35____ Protons, Neutrons, and Electrons Practice Worksheet Fill in the blanks in the following worksheet. Please keep in mind that the isotope represented by each space may NOT be the most common isotope or the one closest in atomic mass to the value on the periodic table. Atomic Symbol B Na Ga Y Cu Tc Pb Yb Ac Mo Tl Fm No Yb Sg Atomic number 5 11 31 39 29 43 82 70 89 42 81 100 102 70 106 Protons Neutrons Electrons 5 11 31 39 29 43 82 70 89 42 81 100 102 70 106 6 13 37 50 35 143 125 102 136 53 125 159 159 100 159 6 11 31 39 29 43 82 70 89 42 18 100 102 70 106 Atomic Mass 11 24 68 89 64 100 207 172 225 95 206 259 261 172 265 Which Atom Is Which? (Hint: White dots are Protons) Every kind of atom has its own unique look. All the atoms of an element have this same look. Here’s a chance for you to look at some atoms and tell what elements they are. Write the name of the element next to each atom. You may need to use the periodic table to help you out. A. Carbon D. Oxygen G. Lithium B. Neon E. Helium H. Nitrogen C. Boron F. Hydrogen I. Beryllium