Geology 1010 Labs
advertisement

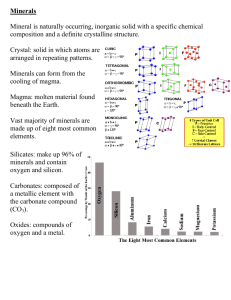
Geology 1010 Labs Geology 1010 – Instructor • • • • • Dr. David McMullin Office: HSH 305 Phone: 585-1276 Email: david.mcmullin@acadiau.ca Office hours: M, W, F, 11:30 – 12:30 Lab assistants • Tuesday: Jackson Malone, Jon Shute • Wednesday: Patrick Henger, Rachel Lewis • Thursday: Margot Aldis Schedule • 10 labs and one final exam (11 in total) • Three sections • You must have permission to switch to another lab section (even for one week) • All lab info on ACORN and lab web site. • All labs are to be handed in at the end of the lab session • Returned to you at the beginning of the next one Things on the lab web page • Lab answer keys – posted after the last lab in the series • Hardness “exercise” • Geological time scale • PowerPoint files of these pre-lab presentations (including this one) • Mineral database (Access) • “Digital learning objects” http://socrates.acadiau.ca/courses/geol/1013/labs/ FOIPOP Act • Graded labs/assignment are considered private property and subject to FOIPOP act • Graded labs with therefore only be handed back in lab Laptops • All lab tests are open book (open laptop) • Bring your textbook to all labs (except field trips) • Bring your laptop to all labs (not necessary on field trips) • Please do not use laptops for non-lab reasons during the lab introductions – Once I’m done you can IM, text, listen to music, whatever, as long as you don’t disturb your neighbours Some personal philosophy • The important thing about any given lab is not to get it done (the end result), but the doing of it (the process). • It’s the process that’s important and one I hope you will bring to new challenges, new opportunities, new situations, new subjects, … for the rest of your life!!! • We tend to lose sight of this because it is the end result that can be graded, but the end result is not what matters most Questions? Minerals • Your textbook defines a mineral as: – an inorganic, – naturally occurring, – crystalline solid (homogeneous solid in which atoms are arranged in a definite order) with a – narrowly defined chemical composition and characteristic physical properties. Minerals are inorganic • Minerals can be formed (secreted) by organic processes but • Materials that require organic processes/activity are not minerals – Sugar is not a mineral, nor is coal, etc. – But teeth are dominantly composed of the mineral apatite (Calcium phosphate) Minerals are naturally occurring • Synthetic analogues of minerals are usually prefaced by the term “synthetic” – E.g. synthetic ruby, synthetic diamond, etc. Minerals are crystalline • Homogeneous • Solid • Ordered atomic arrangement – Quartz is a mineral but glass made from quartz is not (quenched liquid) – water is not a mineral but ice is – Liquid mercury is not a mineral but mercury oxide is Minerals have definite compositions • Many minerals show variation in composition but only within specific limits tightly controlled by their crystalline nature. – E.g. Mg2SiO4 to Fe2SiO4 (olivine) Mineral identification • Physical properties, which are controlled by the crystalline nature • Mineral identification has gone on for thousands of years using very basic tools • Now there are many highly sophisticated methods • But we will be using a few basic tools and your powers of observation Tools dilute HCl hand lens streak plate Cu pennies nail magnet glass plates Physical properties • • • • • • • Lustre Hardness Streak Cleavage/fracture Colour Specific gravity Others Lustre • The way light reflects off the mineral surface. For our purposes we will define only two types of lustre – Metallic (has a metallic sheen or appearance) – Non-metallic • The identification chart (handout) splits the minerals into non-metallic light, nonmetallic dark and metallic Lustre • There are many types of non-metallic lustre. See the display at the back of the lab. Some examples: – Vitreous (glass like) – Porcelainous (like porcelain) – Pearly – Silky – Resinous – Waxy Hardness • The crystalline structure of mineral leads them to have very consistent hardness • Hardness scale puts minerals on a scale of 1 to 10 in terms of their ability to scratch or be scratched by others • Talc has a hardness of 1, diamond is 10 • Hardness scale on Socrates is: http://socrates.acadiau.ca/courses/geol/1013/labs/mohs/mohs.html Hardness • We use a series of simple tools to get an estimate of hardness • Fingernail (approx. 2.5) • Copper penny (approx. 3) • Glass plate and steel tool (both approx. 6) – For our purposes the most important standard is whether it is harder than glass (scratches it) or softer than glass (can be scratched by steel tool) Streak • Streak is the colour of the ground-up mineral • Found by rubbing the mineral on the streak plate • Can only be done with minerals with hardness < 6 – Why? • Many metallic minerals have a diagnostic streak • Most non-metallic minerals have a white streak Cleavage/fracture • The way in which a mineral breaks • Because of the highly ordered atomic structure, many minerals have systematic planes of weaknesses = cleavage • A mineral with one cleavage breaks along a single set of smooth and parallel fracture surfaces which results in a platy appearance One cleavage (platy) Cleavage/fracture • A mineral with two cleavages show two sets of smooth parallel fracture planes at some angle to one-another which commonly results in a rod-like appearance Two cleavages (at right angles) Cleavage/fracture • A mineral with three cleavages shows three sets of parallel planes at angles to one another. Such a mineral will break into – cubes (if the planes are at right angles to one another) or – rhombs (if the angles between the planes are not equal to 90°) Three cleavages Cubic cleavage (e.g., salt) Rhombohedral cleavage (e.g., calcite) Cleavage/fracture • Minerals with 4 or more cleavages are relatively rare and it is difficult to see all the cleavages • Four cleavages produce octahedra (8 sided fragments) • Six cleavages produce dodecahedra (12 sided fragments) Four cleavages Cleavage/fracture • Some minerals do not have cleavage planes and show a random orientation of fracture surfaces • Some minerals without cleavage break with curved surfaces that show ribbing like a shell surface. This is conchoidal fracture Conchoidal fracture ≈60° Colour • Colour is often not a diagnostic property. • Colour variations are most commonly the result of small amounts of impurity • Light coloured non-metallic minerals are more prone to colour variations. Quartz is found in the following colours: Red (carnelian, jasper), pink (rose quartz), purple (amethyst), yellow (citrine), black (smoky quartz), green+red (bloodstone), etc. Colour • Do not confuse the terms “colourless” and “transparent”, they are not the same. – window glass is transparent and colourless – sunglasses are transparent but coloured • Transparent: you can see through • Translucent: allows (some) light through but one cannot see through (e.g., frosted glass) • Transparent objects may coloured or colourless • Translucent and opaque minerals are coloured Specific Gravity • Ratio of the density of the mineral to the density of water • Minerals have specific gravities that vary from about 2.5 to 19 (gold). • Non-metallic light minerals are generally light (SG = 2–3). There are exceptions and such variation is often diagnostic (e.g., barite: SG=4.5) • Non-metallic dark minerals generally have moderate (or average) specific gravity (3–4) • Metallic minerals are generally heavy or very heavy (SG = 5+) Other properties • There are many other properties that minerals can have and they are often diagnostic. A few important ones to watch for: – Magnetism (e.g., magnetite) – Reacts with dilute acid to produce CO2 (e.g. all carbonate minerals to varying degrees) – Tarnish (various metallic minerals) – Fibrous appearance (asbestos) – Iridescence (e.g., plagioclase) – Smell (clay minerals) – Taste (salt) Doing the lab • Fill in the blank spaces in the lab handouts • Many of the spaces are already filled in. Don’t skip these, they are intended to help you learn. • Make sure you understand why each given property is as indicated. For example, #1 is: – Metallic – Has a hardness greater than a penny, less than glass – Has a black streak – Etc. Doing the lab • Use the identification charts (handout) to name the mineral and give its composition. • Look at the examples at the back of the lab. • Ask questions. We will try not to give you flat out answers. Rather we will encourage you to observe and deduce the answers for yourself. • Lab sheet is due at the end of the lab session. One final point… • Enter your observations NOT what you’ve read in the ID chart • E.g., for hardness put “>glass” not “7” (you can determine that something is harder than glass you cannot determine an absolute number) • E.g., for specific gravity, put “H” (heavy, high) not “5.7” • Etc. Questions?