atomic
advertisement

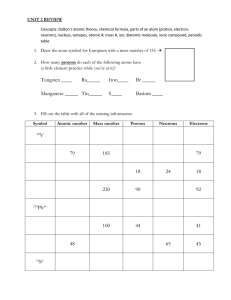
Virtually everything that is, is made up of atoms. Chapter 12, page 361 Balloon and sweeter animation Sub-Particles in the Atom Each element is chemically unique. To understand why they are unique, you need to know the structure of the atom (the smallest particle of an element) and the characteristics of its components. Element = made of one kind of Atom. Li Compounds = made of different atoms chemically bonded in whole number ratios. H2O •Mixtures are physical combinations of elements or compounds with variable composition O2 + H2. Particles or Pure substances Sub-Particles in the Atom Location in the Atom Sub-Particle Mass Charge proton ~1 a.m.u. 1+ in nucleus neutron ~1 a.m.u. 0 in nucleus electron ~0 a.m.u. 1- orbits nucleus atomic.mass.unit= a.m.u. ~ 1.992 ×10−27 kg or 1/12 the mass of C-12 Size of an atom • Atoms are incredibly tiny. • Measured in picometers (10-12 meters) – Hydrogen atom, 32 pm radius • Nucleus tiny compared to atom – Radius of the nucleus near 10-15 m. – Density near 1014 g/cm3 • IF the atom was the size of a stadium, the nucleus would be the size of a marble. California WEB What holds an atom together? • Nuclear Tug-Of-War • • Electrostatic force – like charges repel and un-like charges attract • Strong Nuclear Force – holds nucleons (p+ & n0) • together, very strong nuclear force but over short distances – Stable nuclei are SMALL – Large nuclei tend to be unstable (radioactive) • • Electrostatic Force • – Holds electrons on atom Balloon and sweeter animation Counting Particles in Atoms C Atomic Number = number of protons (p+) = unique for each element 12 6 C 14 6 Mass Number = A C 12 6 Atomic Number = Z Atomic Mass Number = Mass of an atom = number of protons + neutrons = (p+) + (no); (e- do not have mass) California WEB How can I remember how many protons or electrons there are? Just remember the A.P.E. rules! •atomic number = •number of protons = •number of electrons Now, how do I remember how many protons there are? Just remember the M.A.N.! •mass number - •Atomic number = •number of neutrons Neon Ne Protons: 10 Electrons: 10 Neutrons: 10 Atomic Number: 10 Atomic Mass: 20 Phosphorus P Protons: 15 Electrons: 15 Neutrons: 16 Atomic Number: 15 Atomic Mass: 31 Aluminum Al Protons: 13 Electrons: 13 Neutrons: 14 Atomic Number: 13 Atomic Mass: 27 Argon Ar Protons: 18 Electrons: 18 Neutrons: 22 Atomic Number: 18 Atomic Mass: 40 Silicon Si Protons: 14 Electrons: 14 Neutrons: 14 Atomic Number: 14 Atomic Mass: 28 Symbols Contain the symbol of the element, the mass number and the atomic number (top heavy) # protons + # neutrons mass number # protons Mass number Atomic number X Symbols • Find the – Atomic number – Mass number – number of electrons – number of protons – number of neutrons =9 = 19 =9 = 9+ = 10 19 9 F So let’s practice! Symbols Find the – number of protons = 35 – number of neutrons = 45 – number of electrons = 35 – Atomic number = 35 – Mass number = 80 http://www.chem.purdue.edu/gchelp/liquids/bromine.gif 80 35 Br Symbols Find the – number of protons = 11 – number of neutrons = 12 – number of electrons = 11 – Atomic number = 11 – Mass number = 23 23 11 Na Sodium atom Ions To find net charge on an atom, consider the p+ and the e–. cation: a (+) ion anion: a (–) ion -- more p+ than e– -- more e– than p+ atoms -- formed when atoms lose e– -- formed when gain e– - p+ + ++ ion: a charged atom - + + + n0 e- - - - +3 and -4 = -1 - + ++ - p+ + + + n0 e- - +3 and -2 = +1 Symbols Find the – number of protons = 11 – number of neutrons = 12 – number of electrons = 10 – Atomic number = 11 – Mass number = 23 23 11 1+ Na Sodium ion Symbols If an element has an atomic number of 23 and a mass number of 51 what is the – number of protons = 23 – number of neutrons = 28 – number of electrons = 23 – Complete symbol 51 23 V Symbols If an element has 60 protons and 84 neutrons what is the – Atomic number = 60 = 144 – Mass number – number of electrons = 60 – Complete symbol 144 60 Nd Symbols If a neutral atom of an element has 78 electrons and 117 neutrons what is the – Atomic number = 78 = 195 – Mass number – number of protons = 78 – Complete symbol 195 78 Pt Mass Number • mass number= protons + neutrons • always a whole number • NOT on the Periodic Table! + + + Neutron Electrons eNucleus + + e- ee- ee- + Nucleus Carbon-12 Neutrons 6 Protons 6 Electrons 6 Proton Isotopes • Atoms of the same element with different mass numbers. • Nuclear symbol: Mass # 12 Atomic # 6 • Hyphen notation: carbon-12 Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem C Isotopes • Dalton was wrong. • Atoms of the same element can have different numbers of neutrons. • different mass numbers. • called isotopes. Isotopes + + + Neutron Electrons Nucleus + + + Nucleus Carbon-12 Neutrons 6 Protons 6 Electrons 6 Nucleus Proton Proton + + + + Neutron + Electrons + Carbon-14 Neutrons 8 Protons 6 Electrons 6 Nucleus What is an isotope? same number of protons, different numbers of neutrons. B-10 10B 5 Boron-10 Boron-11 B B Protons: 5 Protons: 5 Electrons: 5 Electrons: 5 B-11 Neutrons: 5 Neutrons: 11B 5 Atomic Number: 5 Atomic Number: 5 Atomic Mass:10 Atomic Mass:11 6 What is the average atomic mass of Carbon? Carbon- 12 Carbon- 14 C C Protons: 6 Protons: 6 Electrons: 6 Electrons: 6 Neutrons: 6 Neutrons: 8 Atomic Number: 6 Atomic Number: 6 Atomic Mass: 12 Atomic Mass:14 37 Cl Isotopes 17 • Chlorine-37 – atomic #: 17 – mass #: 37 – # of protons: 17 37 – # of electrons: 17 – # of neutrons: 17 20 Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem Cl Writing Isotopes • Put the mass number after the name of the element • carbon- 12 • carbon -14 • uranium-235 – Or Mass # 12 Atomic # 6 California WEB C Using a periodic table and what you know about atomic number, mass, isotopes, and electrons, fill in the chart: Element Symbol Atomic Number Mass # # of protons # of neutron # of electron 8 8 8 39 Potassium +1 Br 45 30 65 -1 30 Atomic Number = Number of Protons Number of Protons + Number of Neutrons = Atomic Mass Atom (no charge) : Protons = Electrons Ion (cation) : Protons > Electrons charge Ion (anion) : Electrons > Protons Using a periodic table and what you know about atomic number, mass, isotopes, and electrons, fill in the chart: ANSWER KEY Element Symbol Atomic Number Mass # # of protons # of neutron # of electron charge O 8 16 8 8 8 0 Potassium K 19 39 19 20 18 +1 Bromine Br 35 80 35 45 36 -1 Zinc Zn 30 65 30 35 30 0 Oxygen Atomic Number = Number of Protons Number of Protons + Number of Neutrons = Atomic Mass Atom (no charge) : Protons = Electrons Ion (cation) : Protons > Electrons Ion (anion) : Electrons > Protons Isotopes • Because of the existence of isotopes, the mass of a collection of atoms has an average value. • Average mass = ATOMIC WEIGHT = ATOMIC MASS • Boron is 20% B-10 and 80% B-11. – That is, B-11 is 80 percent abundant on earth. • For boron atomic mass = 0.20 (10 amu) + 0.80 (11 amu) = 10.8 amu Average Atomic Mass • massed average of all isotopes on the Periodic Table • round to 2 decimal places Avg. (mass)(%) + (mass)(%) Atomic = 100 Mass Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem Average Atomic Mass • EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 99.76% O-16, 0.04% O-17, and 0.20% O-18. Avg. (16)(99.76) + (17)(0.04) + (18)(0.20) Atomic = 100 Mass Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem = 16.00 amu Average Atomic Mass • EX: Find chlorine’s average atomic mass if approximately 8 of every 10 atoms are chlorine-35 and 2 are chlorine-37. Avg. (35)(8) + (37)(2) Atomic = 10 Mass Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem = 35.40 amu 17 Cl 35.453 • Assume you have only two atoms of chlorine. • One atom has a mass of 35 amu (Cl-35) • The other atom has a mass of 36 amu (Cl-36) • What is the average mass of these two isotopes? 35.5 amu • Looking at the average atomic mass printed on the periodic table...approximately what percentage is Cl-35 and Cl-36? 55% Cl-35 and 45% Cl-36 is a good approximation