Alcohols and Phenols
advertisement

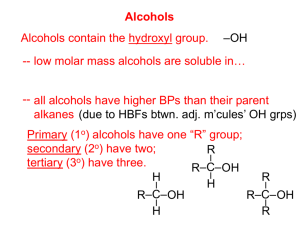
Alcohols and Phenols 1435-1436 2014-2015 1 Alcohols and Phenols Learning Objectives Chapter six concerns alcohols and phenols and by the end of this chapter the student will: know the difference in structure between alcohols and phenols Know the different classes of alcohols Know how to name alcohols and phenols Know the physical properties (solubility, boiling and melting points) Know how hydrogen bonds are formed and its effect on boiling points of alcohols Know the acidic properties of alcohols and phenols know the different methods that can be used to prepare alcohols and phenols. Know the chemical reactions of these compounds ( some reactions are review, others are extensions of the chemistry that will be discussed on chapters 8, 9 & 10 145 Chem. Alcohols and Phenols Structure Of Alcohols and Phenols Alcohols and phenols may be viewed as organic derivatives of water. Water H-O-H R-O-H or PhCH2OH Alcohol Phenol Ph-O-H Alcohols and phenols have a common functional group, the hydroxyl group, —OH. - In alcohols the hydroxyl group is attached to an alkyl group, —R. - In phenols the hydroxyl function is directly attached to benzene ring OH OH 145 Chem. Phenol Benzyl alcohol OH Cyclohexanol 3 Alcohols and Phenols Classification and Nomenclature of Alcohols Alcohols are classified according to the type of the carbon atom connected to the hydroxyl group (carbinol carbon) into: 145 Chem. 4 Alcohols and Phenols NOMENCLATURE OF ALCOHOLS IUPAC Nomenclature Of Alcohols Alcohols are named according to the following rules: 1. Select the longest continuous carbon chain that contains the -OH group. Replace the ending e of the alkane by the suffix –ol 2. If a molecule contains both an -OH group and a C=C or cΞc bond; Choose the chain that include both of them even if this is not the longest chain. The name should include suffixes indicate presence of both the OH group and the unsaturated groups. The OH group takes precedence over the double or triple bonds in getting the lower number. Cyclic alcohols are named using the prefix cyclo-, the carbon bearing the hydroxyl group is assumed to take number 1. 3. The suffix diol is added to the name of the parent hydrocarbon when there are two OH groups. 4. The suffix triol is added when there are three OH groups. 145 Chem. 5 Alcohols and Phenols Common Nomenclature Of Alcohols List the names of alkyl substituents attached to the hydroxyl group, followed by the word alcohol. If two OH groups present on two adjacent carbons these known as glycols Primary alcohols CH3OH CH3CH2OH Common Methyl alcohol Ethyl alcohol IUPAC Methanol Ethanol 145 Chem. CH2=CHCH2OH Allyl alcohol 2-Propen-1-ol 6 Alcohols and Phenols Secondary and tertiary alcohols OH OH CH3 OH Common Isopropyl alcohol IUPAC 145 Chem. 2-Propanol Cyclopentyl alcohol Cyclopentanol Methylcyclohexyl alcohol 1-Methyl-1-cyclohexanol 7 Alcohols and Phenols Examples OH OH OH 1-Phenylmethanol 4-Methyl-2-cyclohexen-1-ol Benzyl alcohol (common name) 5 4 3 2 1 (CH3)2C=CHCH(OH)CH3 4- Methyl-3-penten-2-ol 145 Chem. 2-Phenylethanol OH OH Cl OH 4-Chloro-2-methyl-1-penten-3-o Ethan-1,2-diol Ethelene glycol (common name) 8 Alcohols and Phenols Examples OH OH OH But-3-en-2-ol Pent-4-yn-1-ol 5-Ethyl-hex-5-en-3-ol OH OH CH3 HO 145 Chem. OH OH IUPAC Ethane-1,2-diol Propane-1,2-diol or 1,2-Propanediol Common Ethylene glycol Propylene glycol OH OH Propane-1,2,3-triol or 1,2,3,-Propantriol Glycerol or Glycerin Alcohols and Phenols Physical Properties of Alcohols 1) Boiling Points of Alcohols The boiling points increase with increase in molecular weights. • Alcohols have higher boiling points(b .p.) than alkanes of similar weight this is due to the presence of hydrogen bonds between molecules of alcohols. hydrogen bond OH > OH > OH > Br > Melting & boiling points Increases in this direction 145 Chem. 10 Alcohols and Phenols 2) Solubility Of Alcohols and Phenols Alcohol and water contain hydroxyl group that can form hydrogen bonds. The hydroxyl group is hydrophilic “water loving” The alkyl group of alcohols is hydrophobic “ water hating” As the number of the –OH group in alcohol increases, the solubility in water increases. Diols and triols are more soluble in water than monohydroxyalcohols. The lower alcohols are completely miscible with water As the number of carbons in the alcohol increases, the solubility in water decreases. Homework H2C H C CH2 OH OH IUPAC Nomenclature OH 145 Chem. Commen 11 Alcohols and Phenols 3) Acidic Properties of Alcohols: Alcohols are very weak acids The hydroxyl proton of an alcohol is weakly acidic A strong base can abstract the hydroxyl proton to give an alkoxide ion. R-O-H B R-O CH3CH2OH + B-H ClCH2CH2OH CH3OH 15.5 PKa= 15.9 less acidic 15.3 most acidic Electron-withdrawing group (-I) increase acidity Electron-releasing group (+I) decrease acidity Cl Cl Cl OH > OH > OH > H3CO OH Cl 145 Chem. 12 Alcohols and Phenols Synthesis (Preparation) of Alcohols 1- Hydration From Alkenes H3O+ OH + major 1- Oxidation of Alkenes minor OH OH KMnO4 OH-/H2O 2- From alkyl halide OH cis glycol alcohol,KOH heat H3C C CH2 H (elimination of HX using base gives alkene) CH3CH2CH2Cl dil OHor aq. KOH 145 Chem. CH3CH2CH2OH nucleophilic substitution 13 Alcohols and Phenols 3- Reduction of aldehydes, ketones and carboxylic acids O H LiAlH4 H C C CH3 3 CH3CCH3 or NaBH 4 OH or H2/Pt O LiAlH4 CH3COH 145 Chem. H2 H3C C OH 14 Alcohols and Phenols 4- Addition of Grignard compounds to aldehydes & ketones MgCl O CH3CCH3 + OH H3C C CH3 OMgCl H3C C CH3 H3O+ Tertiary alcohol Ketone Secondary alcohol l Primary alcohol Methanal formaldehyde 145 Chem. 15 Alcohols and Phenols Reaction of Alcohols 1. Dissociation of O-H Bond Reaction as acids (Breaking of oxygen- Hydrogen bond CO ـــــH) A. Formation of Salt (Alkoxide formation) CH3CH2OH Na H2 H3C C ONa Sodium ethoxide B. Formation of Esters H+ H+/ H2O 145 Chem. 16 Alcohols and Phenols 2. Dissociation of C-O Bond Reaction with CــــOH bond cleavage A. Elimination Reactions CH3CH2OH conc.H2SO4 H2C CH2 B. Substitution Reaction CH3CH2OH HCl/ ZnCl2 H3C CH2Cl or SOCl2 or PCl3 or PCl5 145 Chem. 17 Alcohols and Phenols C. Oxidation Reactions CH3CH2OH O H3C CH Cu / or PCC O KMnO4 CH3CH2OH OH CH3CHCH3 H3C Cu / or KMnO4 or PCC , CrO3. HCl PCC = C-OH O H3C C-CH3 Pridinium Chloro chromate N 145 Chem 18 Alcohols and Phenols Nomenclature and acidity of Phenols Phenols (compounds having hydroxyl group directly attached to a benzene ring ) are generally named as derivatives of the simplest member of the family, phenol Acidity : Alcohols and phenols have weak acidic properties. Phenols are much stronger acids than alcohols. Introduction of electron-withdrawing groups, such as NO2 or CN, on the ring increases the acidity of phenols. 145 Chem. 19 Alcohols and Phenols OH OH < OH OH < O2N O2N < < 4-Methylphenol p-Cresol (Common) PH>10 Phenol PH=10 OH NO2 2,4-Dinitrophenol 4-Nitrophenol PH=4 PH=7 OH CHO 145 Chem. Picric acid 2,4,6-Trinitrophenol PH=0.25 OH COOH Cl 2-Hydroxybenzaldehyde o-Hydroxybenzaldehyde Salicylaldehyde NO2 NO2 NO2 CH3 OH 2-Hydroxybenzoic acid o-Hydroxybenzoic acid Salicylic acid NH2 3-Amino-5-chlorophenol 20 Alcohols and Phenols Synthesis of Phenols 145 Chem. 21 Alcohols and Phenols Reactions Phenols Ethers formation O CrO3 / H+ O p-Benzoquinone 145 Chem. 22 Alcohols and Phenols 2. Electrophilic Substitution Reactions 145 Chem. 23 Alcohols and Phenols Home Work KMnO4 OH-/H2O MgCl O HCH + O 145 Chem. H3O+ H2C MgBr H3O+ + 24 Alcohols and Phenols 4) The IUPAC name of A) 4-Ethyl-5-heptyn-3-ol B) 4-Ethyl-5-heptan-3-ol C) 4-Ethyl-5-hepten-3-ol D) 4-Etyl-2-hepten-5-ol 5) The IUPAC name of A) 3-Methyl-1-bromocyclohexanol B) 2-Bromo-3-methylcyclohexanol C) 4-Bromo-2-methylcyclohexanol D) 3-Bromo-1-methylcyclohxanol is: OH is: 6) The most acidic compound is: 145 Chem. 25 Alcohols and Phenols 7) The common name of 2-methyl-2-propanol is: A) Allyl alcohol B) Isopropyl alcohol C) tert-Butyl alcohol D) Benzyl alcohol 8) The following reaction gives A) 4-Ethylphenol B) 2-Ethylphenol C) Ethylphenyl ether D) Ethylphenyl ketone 145 Chem. 26 Alcohols and Phenols 9) The major product for the following reaction is 10) The product of the reaction shown below is 11) Which of the following compounds has the highest boiling point? Alcohols and Phenols Thank You for your kind attention ! Questions? Comments 28