Functional Group Tests
advertisement

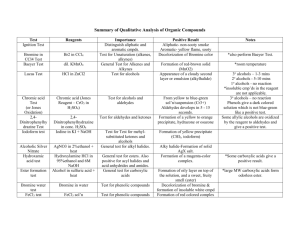
Functional Group Tests 2,4 Dinitrophenylhydrazine Test Shows positive test for: Aldehydes and ketones Reactions: Reacts with the carbonyl group of aldehydes and ketones How to perform the test: Five drops of the compound to be tested are mixed with 5 drops of the dinitrophenylhydrazine reagent (an orange solution) in 2 ml of ethanol and the tube shaken. If no positive test is observed immediately, the mixture should be allowed to stand for 15 minutes. A positive test is indicated by: The formation of a yellow, orange or orange-red precipitate. The KMnO4 Test Shows positive test for: Aldehydes, alkenes and phenols Reactions: Aldehydes are oxidized to carboxylic acids Alkenes are oxidized to glycols Functional Group Tests Monohydroxy phenols do not yield stable products but dihydroxyl phenols can be oxidized to quinones How to perform the test: Three drops of the compound to be tested are dissolved in 2 ml of water or aqueous ethanol. 2% KMnO4 solution (a purple solution) is added drop wise and the solution is shaken. Indications of a positive test: The purple color of the KMnO4 solution disappears and a precipitate of MnO2 is formed. A negative test (left) and a positive test (right) Lucas Test - distinguishes between 1o, 2o, and 3o alcohols Lucas' reagent is a solution of saturated and anhydrous zinc chloride in concentrated hydrochloric acid. This solution is used to classify alcohols of low molecular weight. The reaction is a substitution in which the chloride replaces a hydroxyl group. Lucas Test for Secondary and Tertiary Alcohol Functional Group Tests Alcohols 1-Butanol, 2-Butanol, t-Butyl alcohol. Procedures To 0.2 mL or 0.2 g of the unknown in a test tube add 2 mL of the Lucas reagent at room temperature. Stopper the tube and shake vigorously, then allow the mixture to stand. Note the time required for the formation of the alkyl chloride, which appears as an insoluble layer or emulsion. The Lucas reagent is already prepared for you. Positive test Appearance of a cloudy second layer or emulsion 3o alcohols: immediate to 2-3 minutes 2o alcohols: 5 -10 minutes 1o alcohols: no reaction Functional Group Tests Complete the following table by indicating the functional groups present on each compound and its water solubility (soluble, borderline, insoluble). You can review the structure of each compound by clicking on its name. If you have access to a printer, printing this page would be helpful. compound bp (oC) propanal 49 4-methyl benzaldehyde 204 ethanol 78 1-hexanol 158 t-butanol 82 1-butanol 117 acetone 56 cyclohexene 83 phenol 182 methylcyclohexane 101 functional group water solubility ______________________ ______________ ______________________ ______________ ______________________ ______________ ______________________ ______________ ______________________ ______________ ______________________ ______________ ______________________ ______________ ______________________ ______________ ______________________ ______________ ______________________ ______________ Functional Group Tests pentane 36 ______________________ ______________ Post Questions: Consider the two functional group tests below. a. Tollen’s Test - gives a positive result for aldehydes but not for ketones b. Lucas Test - distinguishes between 1o, 2o, and 3o alcohols.