Citric Acid Cycle & Oxidative Phosphorylation
advertisement

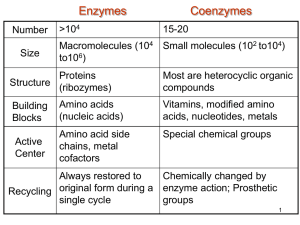
Citric Acid Cycle & Oxidative Phosphorylation • The citric acid cycle, formerly known as the Kreb cycle, begins in the mitochondria as the 2 molecules of pyruvate enter through active transport are further broken down to yield the remaining energy stored in the bonds. • Before the cycle can begin the pyruvate molecules must be converted to Acetyl CoA. – Pyruvate's -COO- (carboxyl group) is removed producing the 1st molecule of CO2 – Acetate (CH3CO-) a 2 carbon compound is produced. – Coenzyme A (from a B vitamin) is added to the molecule via an unstable -S- bond • 2 molecules of CO2 are produced (1 each) • 2 molecules NADH are produced (1 each) which are later used in the citric acid cycle Acetyl CoA Citric Acid Cycle (1-4) • 8 steps make up the citric acid cycle in which the energy in the final 2 carbons are extracted. – Acetyl CoA is attached to an Oxaloacetate (4C) molecule to form citrate (6C). • CoA is released. – Citrate is converted to isocitrate (6C) moving a -OH group (via the removal & addition of water) – a-ketoglutarate (5C) is produced. • CO2 is produced as a carbon is oxidized • NAD+ is reduced to NADH. – Succinyl CoA (4C) is produced as the coenzyme renters into the cycle. • CO2 is produced as a carbon is oxidized • NAD+ is reduced to NADH. Citric Acid Cycle (5-8) – A PO4 group displaces the CoA and is transferred to GDP to form GTP. • GTP then transfers the PO4 to ADP forming ATP and Succinate. – 2 H are transferred to FAD to form FADH2 (energy carrying molecule like NADH) and Fumarate is formed. – H2O is added to form Malate. – Malate is oxidized to form Oxaloacetate • producing a NADH from NAD+ . Citric Acid Summary Glycolysis Acetylation Citric Acid Cycle Oxidative Phosphorylation • The remaining energy stored in the carrier molecules NADH and FADH2 are extracted in the electron transport chain to produce the majority of the ATP in the membranous matrix of the mitochondria. – As the carriers (NAD & FAD) move down the chain they alternate between oxidized and reduced states passing the e- to increasingly electronegative carriers. (exergonic fall of electrons down the chain) 1. Flavoprotein accepts the H from NADH and becomes reduced (NAD+ is oxidized) 2. Fe*S accepts the H and becomes reduced as flavoprotein becomes oxidized 3-5 Ubiquinone is the next molecule (only non protein) which continues the redox chain down through a series of cytochrome molecules The final cytochrome (c) passes the used e- & H+ to 1/2O to form H2O 1. 2. 3. 4. Makes no ATP Manages electrons and removes them to be used in next step Pushes H+ ions to the inner membrane establishing the ion gradient (from NADH & FADH2) Forms H2O at the end of the chain ATP Synthesis • ATP is synthesized by the protein (enzyme) ATP synthase that is ubiquitous in the mitochondrial membrane. – ATP synthase harvests the energy of the proton gradient (proton motive force) to drive a redox reaction and produce ATP via oxidative phosphorylation. • Uses existing ion gradient (H+ ions) • called chemiosmosis – simply put...uses H+ ions stored in the form of a gradient to do cellular work – Movement of ions through the inner channel causes the catalytic head to spin exposing sites for the production of ATP from ADP Cellular Respiration Summary • Glycolysis – changes glucose-6-phosphate to 2 pyruvate molecules • 2 NADH • 2 ATP • Citric Acid Cycle – changes pyruvate into Acetyl CoA then to citrate – cycles ultimately modifying citrate into the beginning molecule Oxaloacetate • 8 NADH (2 from process of changing pyruvate to Acetyl CoA) • 2 FADH2 • 6 CO2 (2 from process of changing pyruvate to Acetyl CoA) • 2ATP • Oxidative Phosphorylation – uses energy stored in the electron carriers and the proton-motive force to product ATP • 32 or 36 ATP (~ 2.5 - 3.3 can be created from ea NADH, 1.5 - 2 from FADH2) – if completely efficient it could create 38 ATP – ~ 40% of the energy in the glucose molecule is ultimately harnessed – DG = -686kcal/mol Variables: 1. the amount of H+ creating the proton-motive force is approximate and variable 2. the ratio of NAD+ or FAD used as carriers (brain uses more FAD, heart & liver uses more NAD) 3. some of the protonmotive force is used to drive the mitochondrion's uptake of substrates type of organism plants use light as energy to drive the production of ATP prokaryotes use the plasma membrane to create the ion gradient and need the gradient for endo & exocitosis as well