4.1 PPT: Electromagnetic Radiation, Quantum, and Electron Excitation
advertisement

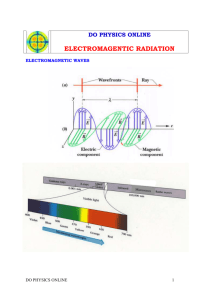
ELECTROMAGNETIC RADIATION AND THE NEW ATOMIC MODEL PROPERTIES OF LIGHT Electromagnetic Radiation A form of energy Is like a wave when it travels through space What are some examples of radiation? PROPERTIES OF LIGHT Electromagnetic Spectrum? All forms of Electromagnetic Radiation are arranged in a spectrum according to their ENERGY! PROPERTIES OF LIGHT Waves are a repeating pattern of motion Wavelength (λ) The length of one repeat of the pattern! Meters, nanometers Frequency (ν) the amount of wavelengths that occur in a unit of time (usually seconds) Waves/sec = Hertz (Hz) PROPERTIES OF LIGHT For electromagnetic radiation… Wavelength and the frequency are related to a wave’s speed c = λν c is in meters per seconds (m/s) PHOTOELECTRIC EFFECT Scientists shined a light on a metal… Some lights caused the metal to eject an electron! Some lights didn’t… They tried increasing the intensity of the lights that didn’t work and that didn’t help either! PHOTOELECTRIC EFFECT Planck realized that while all objects emit electromagnetic radiation (EMR), they don’t emit it continuously like a gradient Instead they emit EMR in little packets called QUANTA. QUANTA A QUANTUM of energy is a specific amount of energy gained or lost by an atom What does this mean? QUANTA Planck’s relationship proposal between quantum energy and electromagnetic wave frequency E = hν E = energy in joules (J) h = Planck’s constant (J•s) = 6.626 x 10-34 J•s QUANTA Einstein proposed: Electromagnetic radiation is both wave-like particle-like Photon – the particle form of a wave of electromagnetic radiation (particle of energy) Ephoton=hv EXCITED STATES AND EMISSION SPECTRA Ground state – relaxed/stable state of matter; lowest energy state When a certain quantum of energy hits an atom an electron can become excited or raised out of the ground state! EXCITED STATES AND EMISSION SPECTRA Emission – when an excited electron relaxes to the ground state, a photon is released. Absorption – when a ground state electron receives the right quanta of energy to become excited. EXCITED STATES AND EMISSION SPECTRA A continuous spectrum from hydrogen – what would happen if hydrogen could be excited by any amount of energy. Line-emission spectrum – the distinct bands of light observed from excited gases EXCITED STATES AND EMISSION SPECTRA E at n = 2 Quantum of energy E at n = 1 absorption relaxation Emission Of photon E2- E1 = hv BOHR’S HYDROGEN ATOM MODEL Bohr thought electrons could only circle the atom in specific pathways, or orbitals. The orbitals were specific energy levels and distances away from the nucleus BOHR’S HYDROGEN ATOM MODEL