Chemistry SLO Review- Extra credit Due: April 23 rd 2015.
advertisement

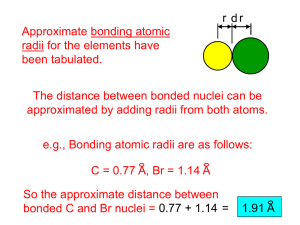
Name: ________________________________ Date: ____________ Period: _____ rd Chemistry SLO Review- Extra credit Due: April 23 2015. Use your text book to complete the following items to help you review for the SLO assessment this week. You do NOT need to copy the question. You MUST show ALL relevant calculations, and answer in complete sentences (if applicable). SCI: Matter and its classifications a. Stated and phases of matter Page 58 # 39, 41, 46, 49, 54 c. Formula writing Page 247 #43, page 282 #65, 66, 70 d. Nomenclature Page 281 # 57, 58, 60, 61 page 282 #67, 68, 69 SC2: chemical composition and chemical reactions a. Reactions Page 347 #43, 52, 57, 58, 67 b. Conservation of mass Page 379 #40, 41 42, 44 c. Moles calculations Page 315 #52, 53, 58, 59, 60, page 380 # 49, 50 d. Limiting reactants Page 379 #47 a, 47 b, page 380 # 51a SCSHh5: computation skills d. significant figures Page 96 # 58, 59, page 250 # 85 e. solving quantitative problems Page 96 #76, 86, 87 SC3: Atomic Theory a. Atomic Structure Page 108 #12, 14, page 122 # 48, page 152 # 90 b. Electron configuration Page 150 #50, 57, 70 c. Atomic identification Page 207 #34, 35, 36 d. Isotopes Page 112 #17, 18 Page 113 # 19, 20 Page 250 # 86, 87 SC4: Periodic table a. Periodic Table origination Page 181 #30, page 182 #48 b. Periodic table trends Page 181 # 36, 38, 39, 42, 43 Continued 1. Calculate the number of electrons, protons and neutrons in all three isotopes of Hydrogen . 2. Calculate the average atomic mass for neon if its abundance in nature is 90.48% neon-20, 0.27% neon21, and 9.25% neon-22. 3. The isotope of an element has 37 protons and 36 electrons, the symbol of the element is 4. The element Sodium has 13 neutrons, then its atomic and mass numbers are 5. Name the electrically charged particles in the atom 6. The conclusions of Rutherford’s atomic model are 7. Pure water, pool water, pond water. Identify the compound, homogeneous and heterogeneous mixture. 8. Isotopes have same number of _________ and different number of ___________ 9. The state of matter that has neither definite shape nor volume is 10. Ne is very unreactive gas, the statement is an example of _________ property. 11. Salt in water is an example of __________ mixture. 12. How do you separate sugar from sugar water solution? 13. Which atomic model is also known as plum pudding model of atom? 14. The five evidences for chemical changes are 15. The number of electrons in 2nd ,3rd and energy levels are 16. The electron configuration of Argon is 17. Write the orbital notation electron configuration of Fluorine. 18. Isotope name: Potassium-39, Identify the nuclear symbol, atomic number, mass number, # protons, # electrons, # neutrons. 19. Calculate the average atomic mass of silver if 13 out of 25 atoms are silver-107 and 12 out of 25 atoms are silver-109. 20. Put the following elements in order from smallest to largest atomic radius and explain why: C, O, Sn, Sr 21. Explain how shielding contributes to the atomic radius trend within a group. 22. Write and formula for sulfur dioxide, dinitrogen tetroxide and sulfur tetrachloride. 23. Put the following elements in order from lowest to highest first ionization energy and explain why: Al, Ar, Cs, Na. 24. Write the name for the following formulas.N2O and SO2 25. Name the following compound below: NH4Br, Fe(NO2)3 and TiBr4. 26. Write the formula for the following compounds: Chromium (VI) sulfate, Vanadium (IV) acetate and Tin (II) nitrate. 27. Name the following compound below: Cu3P2 , SnS2 and GaN 28. Write the formula for the following compounds: Cobalt (II) sulfide, Titanium (II) phosphate and Vanadium (V)oxide 29. Name the following compound below: Pb(SO3)2 Be(HCO3)2 and Mn2(SO4)3 30. Name the following compound below:K2CO3 , N2O5 and CH4 31. Write the formula for the following compounds: Manganese (III) hydroxide, Sodium iodide and Lead (II) phosphide. 32. Name the following compound below: Al(CO3)3 , CrSO4 and SiO2 33. Write the formula for the following compounds: Silver chloride, diphosphorus trioxide and magnesium acetate 34. Name the following compound: GaCl3 CoBr3 and B2H6 35. Calculate the number of moles of Calcium acetate present in 93 g of it. 36. Find the mass of 2.6 moles of Sodium hydroxide. 37. Calculate the volume of 0.4 moles of 𝐶2 𝐻6 38. Balance and indicate the type of reaction. __CaBr2 + ____ H3PO4 ____ Ca3(PO4)2 + ____ HBr 39. Define the periodic table/law. 40. Balance and indicate the type of reaction. __Mg(OH)2 + __Cr2(SO4)3 ____ MgSO4 + ____ Cr(OH)3 41. Metals are found on the __________________ side of the table, while the nonmetals are found on the ___________________ side of the table. 42. Name the member of alkali metals/alkaline earth metal/ metalloid, transition element/ halogen. 43. Name the element that has 6 valence electrons 44. For each of the following, write the correct element that best matches the statement on the right. 45. The element with electron distribution ending in s2p1 46. For each of the following, write the correct element that best matches the statement on the right. 47. Greatest electron affinity among Al, Cl and P is for 48. The largest atomic radius among Ga, Al, Si is for 49. The element with smallest ionization energy among N, P and As is for 50. Largest atomic mass among K, Ca and Sc is for 51. What is the molecular formula of a compound whose empirical formula is 𝐶𝐻2 O and the molar mass is 180 g/mol. 52. Determine the molecular formula of the compound which has C= 50.7%, H = 4.2% , O = 45.1% And the molar mass of the compound is 284 g/mol. 53. Find the number of atoms in 2.8 moles of 𝑁𝐻4 N𝑂3 54. Calculate the percent composition of each element in (𝑁𝐻4 )2 𝐶2 𝑂4 55. How many moles of each reactant is needed to produce 3.6 × 102 grams of C𝐻3 𝑂𝐻 per the equation 𝐶𝑂(𝑔) + 2𝐻2 (g) → C𝐻3 𝑂𝐻(𝑔) . The volume of CO required to react to produce 3.6 × 102 grams of C𝐻3 𝑂𝐻. 56. What element has 1s2 2s2 2p6 3s2 3p6 57. What element has 1s2 2s2 2p6 3s1 58. What element has 1s2 2s2 2p6 3s2 3p6 4s2 3d3 59. What is the general trend in electronegativity as you go across a period? As you go down a group? 60. Identify as element, compound, homogeneous or heterogeneous mixtures. 1. table salt 2.gold 3. the air in DHS 4. carbon 5. Copper 6. Air outside 61. Describe the energy at the atomic level of a solid, liquid, and a gas. 62. Give an example of each: 1. Element 2. Compound 3. Homogeneous mixture 4. Heterogeneous mixture 63. What happens to the temperature of a substance during a phase change? 64. What is the general trend in atomic radius as you go across a period? As you go down a group? 65. Identify as element, compound, homogeneous or heterogeneous mixtures. 1. Mercury 2. Kool-aid 3. fruit salad 4. city air 5. glucose 66. How is an anion and cation radius different from the neutral atoms radius? 67. Give an example of each of the following: a chemical change and a physical change 68. What is the group number for the following groups. 1. Halogens 2. Alkaline Earth Metals 3. Alkali metals 4. Noble gases 5. Transition elements