Chem. 31 * 9/15 Lecture
advertisement

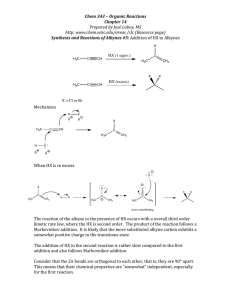
Chem. 31 – 1/26 Lecture What is Quantitative Analysis? (and Why is it important?) • Quantitative Analysis is the determination of a compound’s concentration (or mass or amount) in a sample • Some examples of where a compound’s concentration is important: – level of intoxication from blood alcohol content – determine whether a compound (e.g. F- in drinking water) is beneficial or harmful – risk of having health problems (such as from high LDL concentrations or low HDL concentrations) Roll Call Handouts • Syllabus • Homework Set #1 • Laboratory Report Schedule (discussed more in lab) Typical Lecture Style • Mix of white board or document camera and powerpoint slides • Use white board/document camera for working out detailed problems • Use Powerpoint slides for covering review material (e.g. Chapter 1) or material where having good graphics helps • Powerpoint slides will be made available on website • Announcements given in first few minutes Why is This Course Valuable? • Analysis of chemicals is common in other chemistry classes (e.g. Chem. 25, Chem. 125, Chem. 141, Chem. 161, etc.). • Many of the jobs both within chemical industry/pharmaceutical industry and in applied areas (e.g. environmental service and biotech) involve chemical analysis. Research Projects - some examples of chemical analysis • Quantitation of glycoprotein oligosaccharides (joint with Dr. Peavy) protein Oligosaccharides from protein in Xenopus laevis egg glycoproteins Glucose oligomer standard oligosaccharides 40 20 0 5 7.632 7.723 7.760 7.859 7.940 8.069 8.151 8.349 8.468 8.662 8.969 9.039 9.078 9.125 9.209 9.262 9.472 9.638 9.751 9.859 9.908 10 15 20 25 25.850 24.784 23.834 22.964 22.120 21.249 20.354 19.438 18.502 15.137 15.201 15.258 15.308 15.386 15.655 16.070 16.146 16.206 16.336 16.604 17.009 17.158 17.291 17.556 R 20 10.2 min Chromatogram (each peak = 1 compound) H3C 10 10.4 O R' O H3C H3C 10.6 O R" O O 10.8 10.908 pA 10.742 FID1 B, (Y VONNE\08081301.D) 10.800 O - 10.653 O O 10.585 – biodiesel (on-campus effort to use waste fryer oil to fuel lawn mowers) – synthetic diesel (made from CO + H2) H3C 10.412 O 10.284 O 10.332 • Measurement of new diesel fuels 10.237 1.756 1.824 14.714 180 11.263 11.334 11.386 11.435 11.509 11.669 11.733 11.813 11.864 11.975 12.324 12.356 12.404 12.454 12.527 12.809 12.856 12.965 13.277 13.315 13.369 13.417 13.490 13.810 13.764 14.206 14.259 14.314 14.364 14.439 10.742 9.573 8.269 6.764 5.013 100 4.378 4.407 4.519 4.594 4.643 4.784 4.858 5.142 5.289 5.658 6.008 6.162 6.292 6.423 6.537 6.626 6.706 6.865 6.994 7.164 80 3.244 120 10.237 10.284 10.332 10.412 10.585 10.653 10.800 10.908 60 2.143 pA 2.737 2.768 2.809 2.884 2.974 3.120 3.184 3.317 3.369 3.504 1.634 1.707 1.961 2.040 2.087 2.194 2.267 Research Projects - some examples of chemical analysis R OH O + + + 11 OH R' O OH R" O FID1 B, (Y VONNE\08081301.D) 160 60 140 50 40 30 min Traditional vs. Modern Methods Characteristic Traditional Modern Equipment Glassware and balances (low cost) Instruments (high cost) Precision High Moderate Speed slow fast Sensitivity low high Selectivity minimal Good to great Chapter 1 – Measurements and Titrations No measurement is valuable unless it is given with units and some measure of uncertainty Units – Chapter 1 Uncertainty – Chapters 3 and 4 Units of Measure • Most Basic – Measure Length Mass Time Temperature Amount SI base units (important ones) Unit_____ meter (m) kilogram (kg) (only one with multiplier) second (s) Kelvin (K) Mole (mol) Units of Measure • Directly Derived from Base Units – Volume: cube volume = l3 so units = m3 l – Density = m/V so kg/m3 – Pressure = force(kg·m/s2)/area(m2) = kg/(s2·m) Units of Measure • Other metric units (not directly in SI units) – Density (g/cm3) – Pressure (Pascals or Pa = kg/(s2·m)) • Non-metric units (used commonly) – For pressure 1 atmosphere (atm) = 101300 Pa – English/Other system (not emphasized here) Units of Measure • Metric Name Kilo Centi Milli Micro Nano Multipliers (ones you should know) Abbreviation Multiplier k x103 c x10-2 m x10-3 m x10-6 n x10-9 Analytical chemists like small quantities. An instrument that can detect 1 fg (1 x 10-15 g) is better than an instrument that can detect 1 pg (1 x 10-12 g)