Ch. 1 Power Point notes
advertisement

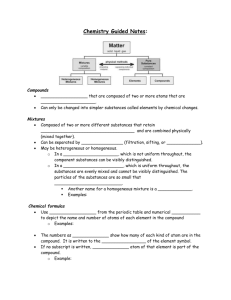
Chemistry is the study about how matter is put together, how atoms combine to form molecules, and how the molecules combine to make up the many kinds of matter around us. Math- the language of Science: 16th century-Science made huge progress when it was discovered that nature can be analyzed and described when scientific findings are expressed mathematically they are easy to verify or disprove be experiment Galileo Galilei (1564-1642) Italian Physicist Francis Bacon (1561-1626) English philosopher Co-Founders of scientific method ~ Extremely effective in gaining, organizing, and applying new knowledge Scientific method – systematic approach to do research, answer questions, and solve problems Recognize a problem Make an educated guess-a hypothesis-about the answer Predict the consequences of the hypothesis Perform experiments to test predictions Formulate the simplest rule that organizes the main ingredients: hypothesis, prediction, and experimental outcomes http://www.youtube.com/watch?v=zrzMhU_4m-g Classification of Matter Matter – anything that has mass and takes up space Mixture- matter that contains two or more different materials Phase- any region with a uniform set of properties Ex: milk, watery part is one phase while fat is the second phase Mixtures are divided into two categories 1. Heterogeneous mixture- a mixture that is composed of more than one phase Ex: milk, granite, sand 2. Homogeneous mixturematerials that consist of only one phase. If you break a piece of homogeneous matter into smaller pieces, each piece will have the same properties as every other small piece Ex: seawater, window glass, and air Substances- homogeneous materials that always have the same composition *Homogeneous mixtures are not pure substances, yet the substances that comprise them are evenly dispersed throughout the mixture Pure Substances are divided into two categories 1. Elementssubstances composed of only one kind of 2. Compoundscomposed of more than one kind of atom atom (elements on the P.T.) Ex: sodium ~ Na, Iron ~ Fe, Neon ~ Ne (made from multiple elements on the P.T.) Ex: water ~ H2O, table salt ~ NaCl Changes in Properties Physical change- same substance remains after a change has taken place *Pounding, pulling, or cutting does not change the chemical character of a substance. Cutting a piece of wood into smaller pieces, tearing paper, dissolving sugar in water, hammering copper into a new shape are all examples of physical change. Physical properties- a description of the behavior of a substance undergoing a physical change two kinds 1. Extensive- depends on the amount of matter present Ex: mass, length, volume 2. Intensive- does not depend on the amount of matter present Ex: density (regardless of size, each sample of a substance has the same density) color, crystalline shape, melting point, boiling point, and refractive index (ability of material to bend light) Chemical change- after a change has taken place a new substance appears Changes that produce a new kind of matter with different properties. Burning, digestion, fermenting all form new substances with different characteristics. Sodium- a silvery, soft metal that reacts vigorously with water (Na) Chlorine- a yellow-green gas that is highlycorrosive and poisonous (Cl) Yet if these two elements are brought together, they combine to form a white crystalline solid. Table salt (NaCl), which neither reacts with water, nor is poisonous. 1. Heat and/or light is produced 2. Production of a gas 3. Color change 4. Formation of a precipitate – a cloudy solid which appears after mixing to clear solutions Intro to the Periodic Table Vertical columns Horizontal rows called groups or families. called periods. Elements in a group have similar chemical & physical properties. Numbered from 1-18 from left to right Elements within a period have properties that change progressively across the table. Metals – (left side) are good conductors of heat and electricity; shiny metallic luster (mostly silver or grayish white); malleable (hammered or rolled into thin sheets); ductile (string into a wire); properties vary according to groups Nonmetals – (right side) elements that are poor conductors of heat and electricity, dull (no luster); some are gases at room temperature. Br is a liquid; C, P, Se, S & I are solid and much more brittle than metals Metalloids – (on the stair-step line) – elements that have characteristics of both metals and nonmetals to a limited degree. Less malleable than metals, but not as brittle as nonmetals; some have luster, some don’t; semiconductors of electricity (used in semi-conducting materials found in computers, calculators, watches, TV’s and radios) Branches of Chemistry Organic Chem – study of carbon – containing compounds Inorganic Chem – all substances not classified as organic (mainly those that do not contain carbon) Physical Chem – the study of properties and changes of matter and their relation to energy Analytical Chem – identification of the components and composition of materials Biochemistry – substances and processes occurring in living things Theoretical Chem – uses math and computers to understand the principles behind observed chemical behavior and to design and predict new compounds