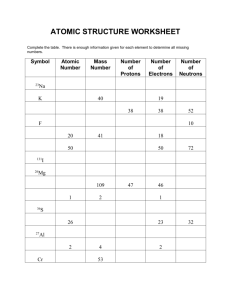
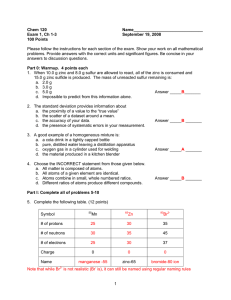
Average Atomic Mass Calculations
advertisement

Unit V: The Mole Average Atomic Mass Calculations WS Learning Target: 1 Directions: Perform the average atomic mass calculations in the space provided. 1. Boron has two naturally occurring isotopes with masses of 10.0129 amu which occupies 19.91 percent and another isotope of 11.0093 amu and occupying 80.09 percent. Calculate the average atomic mass of Boron (The number listed on the periodic table). 2. Bromine has two isotopes with the first having a mass of 78.918336 amu and occupying 50.69% and the second isotope having a mass of 80.916289 amu and occupying 49.31%. What is the average atomic mass of bromine? 3. Verify the atomic mass of chlorine as 35.45 amu knowing that chlorine has two isotopes of the following data: 35 Cl Cl 37 = 34.96885 amu and percent abundance of 75.77% = 36.96590 amu and percent abundance of 24.23% 4. Verify the atomic mass of magnesium as 24.31 amu knowing the following information: 24 Mg = 23.985042 amu and percent abundance of 78.99% Mg = 24.985837 amu and percent abundance of 10.00% 26 Mg = 25.982593 amu and percent abundance of 11.01% 25 5. What is the average atomic mass of lithium if 7.42% exists as 6Li (6.015 g/mol) and 92.58% exists as 7Li (7.016 g/mol)? 6. Neon has two major isotopes, Neon-20 and Neon-22. Out of every 250 neon atoms, 225 will be Neon-20 (19.992 g/mol), and 25 will be Neon-22 (21.991 g/mol). What is the average atomic mass of Neon? 7. What is the atomic mass of Hafnium if out of every 200 atoms, 10 have a mass of 176.00 g/mol, 38 have a mass of 177.00 g/mol, 54 have a mass of 178.00 g/mol, 28 have a mass of 179.00 g/mol, and 70 have a mass of 180.00 g/mol? 8. Without doing any math, are there more Bromine-79 atoms or more Bromine-80 atoms on earth? (Hint: Look at the periodic table.)