- Riverside Preparatory High School
advertisement

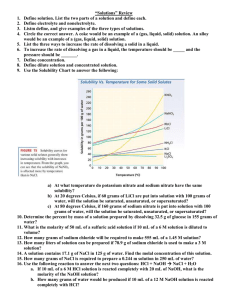
Warm-Up! 1. What is the molarity of an aqueous solution containing 40 g of NaOH in 1.5 L of solution? 2. How many grams of NaCl should be dissolved in 100 L of salt water to have 0.01 M solution? Solution Concentrations Teach us! 1. Density 1. What is the density of 0.5 L diet coke that has 0.05 g of sugar in it? 2. How many grams of sugar are dissolved in a 0.355 L can of regular coke with the density of 110 g/L? 2. Molarity (M) 1. 0.1 L intravenous (IV) solution contains 5.1 g of glucose (C6H12O6). What is the molarity of this solution? The molar mass of glucose is 180.18 g/mol. 2. How many grams of NaOH are needed to produce 0.25 L of a 3M solution? 3. Molality (m) 1. What is the molality of a solution containing 50 g of NaCl dissolved in 1 kg of water? 2. How many grams of NaOH are needed in 0.5 kg of water to make a 1 m solution? 4. ppm (parts per million) 1. 650 g of hard water contains 0.101 g of Calcium. What is this concentration in parts per million? 2. 870 g of river water contains 0.023 g of Cadmium. Express the concentration of Cadmium in parts per million. 5. % by mass 1. In order to maintain a Sodium Chloride (NaCl) concentration similar to ocean water, an aquarium must contain 3.6 g NaCl per 100 g of water. What is the percent by mass of NaCl in the solution? 2. You have 1500 g of a bleach solution. The percent by mass of the solute, Sodium Hypocholorite (NaOCl) is 3.62%. How many grams of NaOCl are in the solution? 6. % by volume 1. What is the percent by volume of ethanol in a solution that contains 0.035L of ethanol dissolved in 0.155 L of water? 2. If 0.018 L of methanol is used to make an aqueous solution that is 15% by volume, how many L of solution is produced?