Molecular Geometry Notes
advertisement

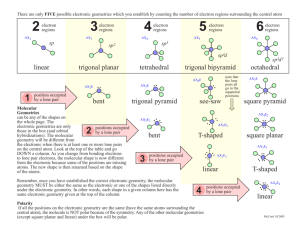
Molecular Geometry Notes February 3, 2014 • You will be able to describe the molecular geometry of a molecule. • Correct HW 6B • A note on nomenclature • Notes • Cool video of the day: Check out THIS ionic crystal lattice!! Binary Nomenclature • First element by name (with prefix, ONLY if there is more than one atom) • Second element as an “-ide”, with prefix • Does not work for molecules with more than 2 atoms • • • • • • • • • • CO NO SF2 SO2 NO2 NCl3 CCl4 H2O N2F6 N2O5 Carbon monoxide Nitrogen monoxide Sulfur difluoride Sulfur dioxide Nitrogen dioxide Nitrogen tricloride Carbon tetrachloride Dihydrogen monoxide Dinitrogen hexafluoride Dinitrogen pentoxide Molecular Geometry • Applies to any Lewis diagram. (Involved in covalent bonding) VSEPR Theory • Valence Shell Electron Pair Repulsion • Each bonding group or lone pair of electrons spreads out as far as possible from every other bonding group or lone pair of electrons. • Think 3D! Electron Pair vs. Molecular Geometry • Electron pair geometry describes both bonds AND lone pairs located around the central atom. • Molecular geometry describes only the bonds located around the central atom. • Molecular geometry is a subset of electron pair geometry. Practice How many bonding groups are around the central atom? 4 How many lone pairs around the central atom? 0 Practice How many bonding groups are around the central atom? 3 How many lone pairs are around the central atom? 1 Practice How many bonding groups are around the central atom? 3 How many lone pairs are around the central atom? 0 Practice How many bonding groups are around the central atom? 2 How many lone pairs are around the central atom? 0 Electron Pair Geometry: Overview (Not in notes) • The shape of the molecule including both bonds and lone pairs • Linear – 2 e- groups from central atom • Trigonal Planar – 3 e- groups from center • Tetrahedral – 4 e- groups from center Any of these lobes (electron groups) can be either lone pairs or covalent bonds REMEMBER • ELECTRON PAIR GEOMETRY and MOLECULAR GEOMETRY are not the same thing. – EPG describes all electron groups (including lone pairs) – MG describes only bonding groups 2 Electron Groups • Electron pair geometry: Linear – 2 bonding groups – 0 lone pairs – Molecular geometry: Linear – Example: CO2 • 2 bonding groups • 0 lone pairs Double and triple bonds count as just ONE bonding group!!! 3 Electron Groups • Electron pair geometry: Trigonal Planar – 3 bonding groups – 0 lone pairs – Molecular geometry: Trigonal planar – Example: NO3This Lewis structure also shows the molecular shape (trigonal planar). Double and triple bonds count as just ONE bonding group 3 Electron Groups • Electron pair geometry: Trigonal Planar – 2 bonding groups – 1 lone pair – Molecular geometry: Bent – Example: NO2Lone pair is one of the three electron groups around the central atom Double and triple bonds count as just ONE bonding group 4 Electron Groups • Electron pair geometry: Tetrahedral – 4 bonding – 0 lone pairs – Molecular geometry: Tetrahedral – Example: CH4 This Lewis structure does NOT show the tetrahedral molecular shape. It DOES show the four electron groups. 4 Electron Groups • Electron pair geometry: Tetrahedral – 3 bonding – 1 lone pair – Molecular geometry: Trigonal Pyramidal – Example: NH3 This Lewis structure does NOT show the molecular shape. It DOES show the four electron groups. Lone pair is one of the four electron groups around the central atom 4 Electron Groups • Electron pair geometry: Tetrahedral – 2 bonding groups – 2 lone pairs – Molecular geometry: Bent/Angular – Example: H2O This Lewis structure shows both the molecular shape and the four electron groups Lone pairs are two of the four electron groups around the central atom Molecular Geometry: Overview • The shape of the molecule including just bonds between atoms. • 2 e- groups from central atom: – If 2 bonds/0 lone pairs: Linear • 3 e- groups from central atom: – 3 bonds/0 lone pairs: Trigonal planar – 2 bonds/1 lone pair: Bent • 4 e- groups from central atom: – 4 bonds/0 lone pairs: Tetrahedral – 3 bonds/1 lone pair: Trigonal pyramidal – 2 bonds/2 lone pairs: Bent Sketches of Molecular Geometry • Sketches (on homework, quizzes, and tests) should show the shape of the molecule in three dimensions: Exceptions to the Octet Rule (SF , BrF , XeF , IF ) (Beyond the scope of this class) 6 5 4 7