Organic Chemistry - Liberty Union High School District
advertisement

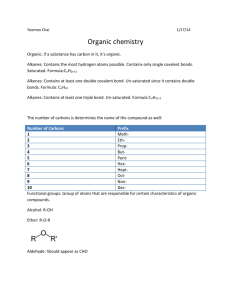
Organic Chemistry Long-Chained Carbon Molecules and Basic Functional Groups Standards 10. The bonding characteristics of carbon allow the formation of many different organic molecules of varied sizes, shapes, and chemical properties and provide the biochemical basis of life. As a basis for understanding this concept: 10. b. Students know the bonding characteristics of carbon that result in the formation of a large variety of structures ranging from simple hydrocarbons to complex polymers and biological molecules. 10. d.* Students know the system for naming the ten simplest linear hydrocarbons and isomers that contain single bonds, simple hydrocarbons with double and triple bonds, and simple molecules that contain a benzene ring. 10. e.* Students know how to identify the functional groups that form the basis of alcohols, ketones, ethers, amines, esters, aldehydes, and organic acids. 10. f.* Students know the R-group structure of amino acids and know how they combine to form the polypeptide backbone structure of proteins. Lewis Dot Shorthand • Each corner (or endpoint) is a carbon atom. Carbon normally has 4 bonds. • Single bonds with hydrogen atoms are not shown they are implied. H H H C H C H H C H H Lewis Dot Shorthand • Each corner (or endpoint) is a carbon atom. Carbon normally has 4 bonds. • Single bonds with hydrogen atoms are not shown they are implied. H H H C H C H H C H O H OH Lewis Dot Shorthand • Each corner (or endpoint) is a carbon atom. Carbon normally has 4 bonds. • Single bonds with hydrogen atoms are not shown they are implied. H H H C H C Cl C Cl H Lewis Dot Shorthand • Each corner (or endpoint) is a carbon atom. Carbon normally has 4 bonds. • Single bonds with hydrogen atoms are not shown they are implied. H H H H C H C H C C H H H C H C H H Lewis Dot Shorthand • Each corner (or endpoint) is a carbon atom. Carbon normally has 4 bonds. • Single bonds with hydrogen atoms are not shown they are implied. H H C H C C C H C H C H Organic Chemistry Prefixes How many Carbons does it have in a row? (the longest continual chain) # of Carbons 1 2 3 4 5 Prefix methethpropbutpent- # of Carbons Prefix 6 hex7 hept8 oct9 non10 dec- Alkanes All hydrogen and carbons with only single bonds End with “-ane” suffix. H H C 1 H H methane Alkanes All hydrogen and carbons with only single bonds End with “-ane” suffix. H H 1 H C C H 2 H 3 C H H 1 3 propane H propane 2 Alkyl- Groups Sometimes there is an extra branch coming off of the main carbon chain. # of Carbons Name 1 methyl 2 ethyl 3 propyl 4 butyl 5 pentyl 1 2 3 5 4 3-methyl pentane The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkyl- Groups Sometimes there is an extra branch coming off of the main carbon chain. # of Carbons Name 1 methyl 2 ethyl 3 propyl 4 butyl 5 pentyl 1 2 3 5 4 3-ethyl pentane The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkyl- Groups Sometimes there is an extra branch coming off of the main carbon chain. # of Carbons Name 1 methyl 2 ethyl 3 propyl 4 butyl 5 pentyl 1 2 3 5 4 2-methyl 3-ethyl pentane The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkyl- Groups Sometimes there is an extra branch coming off of the main carbon chain. # of Carbons Name 1 methyl 2 ethyl 3 propyl 4 butyl 5 pentyl 1 2 3 5 4 2, 3-dimethyl pentane The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkyl- Groups Sometimes there is an extra branch coming off of the main carbon chain. # of Carbons Name 1 methyl 2 ethyl 3 propyl 4 butyl 5 pentyl 1 2 3 5 4 2, 3, 4-trimethyl pentane The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Practice Color the longest continuous carbon chain (turning is okay, not splitting). Do not color the hydrogens. H H C H H H C C H H H C H H C C H H C C H H 5-propyl decane H H C H H H C H C H H H H C H H C H H Practice Color the longest continuous carbon chain (turning is okay, not splitting). Do not color the hydrogens. H H C H H H C C H H H C H H C C H H C C H H 5-propyl decane H H C H H H C H C H H H H C H H C H H Practice Color the longest continuous carbon chain (turning is okay, not splitting). Do not color the hydrogens. H H C H H H C C H H H C H H C C H H C C H H 5-propyl decane H H C H H H C H C H H H H C H H C H H Practice Color the longest continuous carbon chain (turning is okay, not splitting). Do not color the hydrogens. H H10C H H H 9C 8 C H H H Off #5 Carbon H 7C 6 C 5 C H H 4C H 3C H 10 carbons on main chain = decane H H H C H H H 2C H 1C H H H H C H H C H H 3 carbons on branch = propyl 5-propyl decane Alkenes All hydrogen and carbons with single bonds and some carbon-carbon double bonds. End with “-ene” suffix. H 1 C H ethene 2 C H H 2 1 ethene Alkenes All hydrogen and carbons with single bonds and some carbon-carbon double bonds. End with “-ene” suffix. H H H 1 C C 2 H 3 C H H 2-butene 4 C H 2 H 1 3 4 2-butene The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkenes All hydrogen and carbons with single bonds and some carbon-carbon double bonds. End with “-ene” suffix. H H 1 C H C 2 H 3 C H 1-butene H 4 C H 2 H 1 3 4 1-butene The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkenes All hydrogen and carbons with single bonds and some carbon-carbon double bonds. End with “-ene” suffix. 1 2 5 3 4 6 2, 4-hexadiene The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkenes All hydrogen and carbons with single bonds and some carbon-carbon double bonds. End with “-ene” suffix. 1 3 2 5 4 6 1, 3-hexadiene The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkenes with Alkyl Groups All hydrogen and carbons with single bonds and some carbon-carbon double bonds. End with “-ene” suffix. 1 3 2 5 4 6 2-methyl 1, 3-hexadiene The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkynes All hydrogen and carbons with single bonds and some carbon-carbon triple bonds. End with “-yne” suffix. H 1 C 2 C ethyne H 1 2 ethyne Alkynes All hydrogen and carbons with single bonds and some carbon-carbon triple bonds. End with “-yne” suffix. H H 1 C 2 C C 3 propyne H 1 2 H propyne 3 Alkynes All hydrogen and carbons with single bonds and some carbon-carbon triple bonds. End with “-yne” suffix. 5 3 2 4 1 1 - pentyne The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alkynes with Alkyl Groups Sometimes there is an extra branch coming off of the main carbon chain. 5 3 2 4 1 3-methyl 1-pentyne The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alcohols Just like an alkane, except they have an –OH functional group hanging somewhere. End with “-anol” suffix. H H H 1 O C C H 2 H 3 C HO H 2 1 3 1-propanol H H 1-propanol The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Alcohols Just like an alkane, except they have an –OH functional group hanging somewhere. H End with “-anol” suffix. O H 1 H C C H 2 H 3 C H H 2-propanol HO H 2 1 3 2-propanol The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Ketones They have an =O functional group hanging somewhere in the middle of the carbon chain. End with “-anone” suffix. O H 1 H C H O C H 2 3 C acetone H 1 H 2 3 acetone Ketones They have an =O functional group hanging somewhere in the middle of the carbon chain. End with “-anone” suffix. O 1 2 3 4 5 3-pentanone The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Ketones They have an =O functional group hanging somewhere in the middle of the carbon chain. End with “-anone” suffix. 1 3 2 4 5 O 2-pentanone The number comes from the carbon (with the lowest number) that has the feature (aka functional group). Aldehydes They have an =O functional group hanging at the end of the carbon chain. End with “-anal” suffix. H 1 C H H H 2 C O O C H propanal 1 2 3 H 3 propanal H R- Groups R- Groups are a generic way of saying that there is some kind of larger carbon chain attached (even though we won’t specify what it is). O O O C C C H R aldehyde R OH R’ R carboxalic ketone acid H He Li Be B C N O F Na Mg Al Si P S Cl Ar K Ca Ne Br Kr I Xe H He Li Be B C N O F Na Mg Al Si P S Cl Ar K Ca Ne Br Kr I Xe 4 e– in valence shell Organic Chemistry Prefixes How many Carbons does it have in a row? (the longest continual chain) # of Carbons Prefix # of Carbons Prefix 1 meth6 hex2 eth7 hept3 prop8 oct4 but9 non5 pent10 dec- Finish • • • • • How many Cs and Hs in benzene, show abbreviated hexagon. (only for alkanes?) Number of carbon prefixes: 1-10 (done) Meth-, eth-, prop-, but-, pent-, hex-, hept-, oct-, non-, dec- (done) Alkanes, alkene, alkynes, alcohols, ethers?, ketones, aldehydes, amino acids (nope?) What do R groups stand for H H H H C H C H C C H H H C H C H H