Organic Chemistry Notes
advertisement

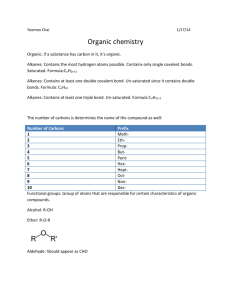
Organic Chemistry Notes Organic chemistry is concerned with __carbon______-containing molecules. Many of the ____compounds_______ found in all ____living_____ things contain carbon! So let’s talk carbon: Atomic symbol: ___C________ Atomic Number: ___6_______ Atomic Mass: ______12 anu__ # protons: ______6__________ # neutrons: ______6_________ # electrons: _______6________ # valence electrons: __4______ Bohr Model: Since carbon has __4_____ valence electrons, it needs ___4___ more to complete its valence shell. Each carbon atom will form __4____ __covalent________ bonds with other atoms. Carbon will form these bonds with: Other ___carbons______, __H_______, the halogens (___F___, _Cl____, ___Br___, ___I____, ___At____) or ___O___, __N_____, __S_____, or ____P___ Hydrocarbons These compounds contain only ___carbon_______ and _____hydrogen______. There are 2 different categories of hydrocarbons: ____Aliphatic______, which has a ____simple____ structure, C’s arranged in a ___chain_________. ___Aromatic_________, which has a ____ring______ structure an d contains ___double_____ bonds between some C atoms. Aliphatic Hydrocarbons There are ____four_____ categories of aliphatic hydrocarbons. 1. ___Alkanes_______: only ___single_______ bonds between carbon atoms 2. ___Alkenes_______: contain one or more ___double______ bonds between carbon atoms 3. ___Alkynes_______: contain one or more _____triple_ bonds between carbon atoms 4. ____Cyclic______: carbon atoms are joined together in a ____ring________ Alkanes The simplest of all aliphatic hydrocarbons, they contain only ___single____ bonds between carbons. Example: Alkanes have the ___maximum_________ number of hydrogen possible to complete carbon’s __4____ bonds. Hydrocarbons with only __single______ bonds and the maximum number of H’s are considered to be _____saturated_________. *** Remember saturated and unsaturated fats? Saturated fats have the ___maximum___________ number of ___H’s____ possible. Naming Alkanes Alkanes are named for the __number_______ of carbons ___bonded________ together. The ____longest_______ chain is called the ___parent________ chain. To name an alkane, _____count____ the number of carbons in the chain, add the correct ____prefix_____, and the name ends in __”—ane”______. Naming Organic Compounds # of C in a Chain Prefix 1 Meth- 2 Eth- 3 Prop- 4 But- 5 Pent- 6 Hex- 7 Hept- 8 Oct- 9 Non- 10 Dec- Example: # of carbons in parent chain: ___5____ Correct prefix: ______pent-_________ Prefix + “—ane” = ______pentane___________ NOW YOU TRY!!!! # carbons in chain: ___3______ = ____propane_________ # carbons in chain: ____4_____ = ______butane________ # carbons in chain: ____2_____ = ______ethane________