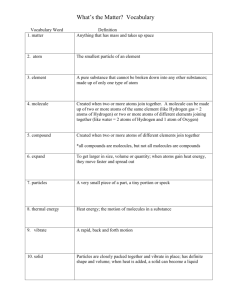
Molecules notes
advertisement

• A molecule is a group of 2 or more atoms linked together chemically. Ex: H20, CH4, O2 ,H2 ,O3 • A compound is a molecule that contains 2 or more different atoms. Ex: CH3COOH (vinegar) Interesting molecules • The caffeine molecule • Lactic acid (in milk) • Adrenalin! Sugar C12H22O11 Vitamin C Fat molecule Are you hungry? Properties of molecules • A molecule has different properties than the atoms it’s composed of. Example: Sodium, Na, is a reactive metal and chlorine (Cl is a toxic gas!) But the two atoms linked together give table salt, that we eat. NaCl Nomenclature and chemical formula • Chemical formula: it is the name of a molecule using symbols. Ex: NaCl, H2O, l, etc. H2O Subscript: indicates how many atoms are present. Affects the atom on the left of it. There are 2 atoms of H and 1 atom of O H2SO4 has 2 atoms of Hydrogen, 1 atom of sulfur, and 4 atoms of oxygen. Let’s practice! Tell how many atoms of each kind there are in the following molecules: a) MgCl2 b) Al2O3 c) H3PO4 d) NH3 e) HCN f) C2H2 • Nomenclature: is the scientific name we use to name a molecule. • The common name of NaCl is table salt. The scientific name is sodium chloride. • The common name of CH3COOH is vinegar! The scientific name is acetic acid.