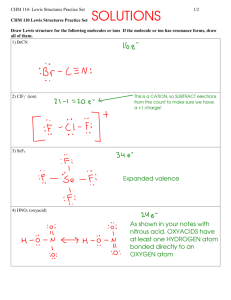
Temperature and Reaction Rate
advertisement

CHM 101/102 Temperature and Reaction Rate General Chemistry 101/102 Laboratory Manual University of North Carolina at Wilmington Laboratory Manual CHM 101/102 Temperature and Reaction Rate • Purpose To study the effect of temperature on the reaction rate and the reaction rate constant (k) of a chemical reaction and to determine the value of the activation energy (Ea) for the reaction. • Safety Considerations Wash your hands thoroughly after handling chemical reagents and before leaving the lab. All chemical wastes should be placed in the labeled jar in the hood. Laboratory Manual CHM 101/102 Temperature and Reaction Rate • Background The chemical reaction being studied in this experiment is: 6 I- + BrO3- + 6 H+ 3 I2 + Br - + 3 H2O bromate ion In the previous experiment, you determined the rate law for this reaction to be: Rate = k [I-] [BrO3-] [H+]2 The value of the rate constant (k) depends on the temperature. The relationship between rate constant and temperature is given by the Arrhenius Equation k = Ae-Ea/RT Laboratory Manual CHM 101/102 Temperature and Reaction Rate • Background rate constant activation energy k = Ae-Ea/RT frequency factor temperature (K) Idea Gas constant (8.31 J/K·mol) Taking the log of both sides of the Arrhenius Equation transforms it into a linear equation: ln k = - Ea + ln A RT Laboratory Manual CHM 101/102 Temperature and Reaction Rate • Background The activation energy for a reaction can be found by measuring the value of k at several different temperatures and then plotting ln k vs. 1/T ln A Laboratory Manual CHM 101/102 Temperature and Reaction Rate • Procedure Each trial is performed at a different temperature (approximately 0 ºC, 10 ºC, 25 ºC, and 40 ºC). Measure the quantities shown in Table 1 into flasks labeled “A” and “B”. B Add 4 drops of starch to flask “B” Allow both flasks to reach the desired temperature before combining them into flask “A”. Keep flask “A” at the desired temperature during the reaction. Record the time it takes for the blue color to appear. Laboratory Manual CHM 101/102 Temperature and Reaction Rate • Calculations A sample calculation for k is shown below. -5 M 3.3 x 10 Rate = = k [I-] [BrO3-] [H+]2 Dt 3.3 x 10-5 M k= Dt [I-][BrO3-][H+]2 3.3 x 10-5 M k= (180 s)(0.0020 M)(0.0080 M)(.020 M)2 k = 29 L3/mol3·s Laboratory Manual CHM 101/102 Temperature and Reaction Rate • Calculations Enter your data into an Excel spreadsheet as shown below and use GraphData to determine the slope of the line. Laboratory Manual