Naming_Ionic_Compounds
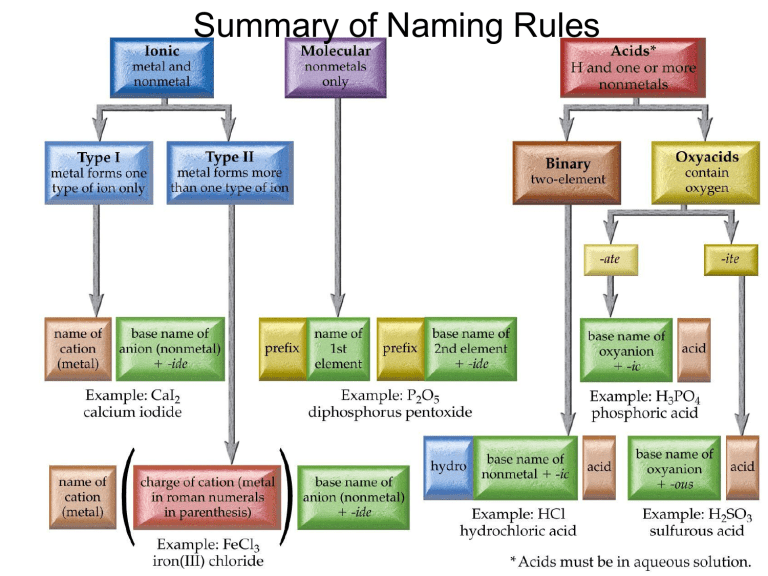
Summary of Naming Rules
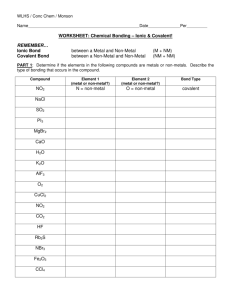
Naming Ionic Compounds
Rule number #1: Make sure you actually have an ionic compound or not!
An Ionic Compound must have a METAL
(except in the case of ammonium NH
4
+1 ) and a NON-METAL
Example:
CrO consists of chromium and
Oxygen
metal non-metal
Rule number #2: Write out the correct names for the parts of the ionic compound
Example: Cr is NOT Chlorine or Carbon or Cryptonite! It is chromium!
CrO chromium and oxygen
Rule number #3:Put an –ide on the ending of the non-metal if it is just an monatomic anion such as Cl or Br or F.
CrO
Example: Chromium Ox ide
Rule number #4: Figure out if you need a Roman numeral in the name.
Cr definitely can have more than one charge:
Cr +2 or Cr +3
CrO
so a roman numeral is needed
Cr +2 or Cr +3
Rule number #3: Figure out what the
Roman numeral should be.
?
Cr O
-2 chromium oxide
Oxygen ALWAYS has a charge of -2
Therefore the Cr has to be +2 the formula is:
Chromium (II) Oxide
Click on this picture to go to a website which will help you find ionic formulas