batteries
advertisement

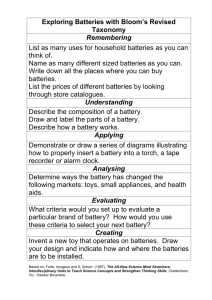
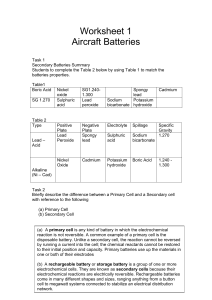
How to power your smartphone for a week! What is a battery? • A device that stores chemical energy in its active materials and converts it, on demand, into electrical energy by means of an electrochemical reaction - + Electron flow Ion flow Cathode • Batteries are made up of one or more basic electrochemical units called cells. Anode – Electrochemical reaction is a chemical reaction involving the transfer of electrons – Cells are usually connected in series to increase the voltage. 2 Presentation name Sentech Electrolyte Types of batteries • Primary batteries are used once and thrown away, like the alkaline batteries used in portable CD players – Electrochemical reactions are not reversible and active materials cannot be restored to their original state. Primary 34% Secondary 66% • Secondary (or rechargeable) batteries can be used many times, like the battery in cell phones and laptop computers 3 Presentation name Sentech – The electrochemical reactions are reversible, and the active materials can be restored to their original chemical composition. Widely used rechargeable batteries Market share • Lead acid (30-40 Wh/kg, 70-92% eff., 2V) – Over 100 years old, and still the most widely used rechargeable battery in the world. 51% • Nickel metal hydride (30-80 Wh/kg, 66% eff., 1.2 V) – A high power battery chemistry similar to nickel cadmium, introduced in the 1980s. It is environmentally friendly, contains no toxic cadmium, and is replacing NiCd in many applications. 8% • Lithium ion (160 Wh/kg, 99.9% eff., 3.6-3.7V) The newest and fastest growing rechargeable battery technology. – Theoretical capacity: 150-275mAh/g Main concerns: large volumes, liquid technology, leakage, and toxicity 4 Presentation name 31% Sentech, Wikipedia – Real batteries 5 Presentation name Research batteries at ORNL 6 Presentation name Actual technology and materials Li-Air Li-S New chemistries Pb Acid Spiral Wound Emergency Aircraft 10000 Where we need to Aircraft go FCS Weapons 1000 Vehicle Mobility SLI 100 Commercial Hybrids BA5590 Soldier Apps Soldier Underwater 10 Where we are today 1 0 20 40 60 80 100 120 140 160 180 Specific Energy, Wh/kg 7 Presentation name BA5347 & BA5123 Soldier Apps © Ming Au, SRNL Specific Power, W/kg 100000 200 220 240 Cell potential © pinkmonkey.com 8 Presentation name © Maxim Integrated Products Cell capacity It C F N Ae n n zF C M LiC 6 Li 1e C6 Li22 Si5 22 Li 22e Si5 9 Presentation name z 1; M (C ) 6 12.01 C 0.372 g mol Ah g z 22; M ( Si ) 5 28.09 C 4.200 Ah g g mol Cell energy • Capacity x voltage Overcharge • Cut off voltage is controlling available energy but also limiting access for safe operation Overdischarge 10 Presentation name Lithium ion battery principle Discharge reaction: 11 Presentation name Cathode Anode Li+ + e- + MeO2 → LiMeO2 LiC6 → Li+ + e- + C6 Development targets and accomplishments Plug-in Hybrid electric vehicles (PHEV) Hybrid electric vehicles (HEV) • D. Howell, Energy Storage Research and Development, Annual Progress Report 2006 • FreedomCAR and Fuel Partnership and United States Advanced Battery Consortium, Electrochemical Energy Storage Technical Team Technology Development Roadmap (Southfield, MI: USCAR, 2006). 12 Presentation name Fruit battery – Natural galvanic element 13 Presentation name Have fun and stay safe! 14 Presentation name