1 Ionic compounds u7 1112 cp
advertisement

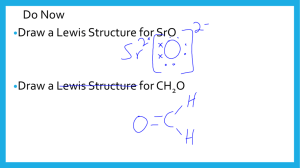
REVIEW CHAPTER 7 IF NEEDED AND COMPLETE #S 27, 29, 35, 38, AND 40 ON PAGE 214 Day 1 4-4 ______ compounds break apart into ______ when dissolved in water and conduct electricity. How do we name and represent different substances? Day 6 4-23 1.Lewis Dot structure for francium bonded to chlorine: 2. Ionic bonds most commonly form between??? 3. Where would you find elements that form cations on the periodic table? Ionic bonds – Chemical bonds that result from the electrical attraction between large numbers of cations and anions Cation – ? Anion – ? Ionic bonds most commonly form between …? Ionic bonds are made up of charged pieces (ions) … Ions can be made up of single atoms or groups of atoms bonded together (polyatomic ions) Binary ionic compound – ionic compound consisting of only 2 elements (monatomic ions) Naming: Name the cation and add an ide ending to the anion Examples: sodium and chlorine form… sodium chloride magnesium and oxygen form… magnesium oxide Bromine bonds with sodium Sodium bromide Cesium bonds with oxygen Cesium oxide Silver bonds with fluorine Silver fluoride chlorine bonds with lithium Lithium chloride magnesium bonds with sulfur Magnesium sulfide aluminum bonds with oxygen Aluminum oxide Binary ionic compound = ? Binary ionic compound – ionic compound consisting of only 2 elements (monatomic ions) REVIEW CHAPTER 7 IF NEEDED AND COMPLETE #S 35, 38, AND 40 ON PAGE 214 Problem… names do not directly indicate ratio of ions… Answer… empirical formula Li2O NaCl MgF2 Determining formulas – Crisscross method Determining formulas – Crisscross method 1. determine charges 2. Take away the +/- and crisscross them to determine subscripts Determine the empirical formula for an ionic compound containing… potassium and fluorine calcium and fluorine cesium and sulfur aluminum and oxygen REVIEW CHAPTER 7 IF NEEDED AND COMPLETE # 40 ON PAGE 214 Read / review section 9.1 and complete #s 3-8 on page 269 4-24 day 1 1. Test Unit 6 = ___% 2. I prepared by… 3. Next time I will prepare by… 4. Average test grade so far = ____% New Goal = ____ you must improve your average by at least 2 percentage points Name the compounds formed when the following elements bond: 1. Calcium and fluorine 2. Chlorine and strontium Day 1 4-24 3. Silver and oxygen Write the formulas for the following compounds: 1. Lithium bromide 2. Barium chloride 3. Sodium oxide 1.Get out the new notes. What is a binary ionic compound? Throwing in the transition metals: Use roman numerals if more than one charge is possible Roman numeral = charge, if none use sheet, then same as binary ionic compounds The weird guys… Throwing in the transition metals: FeO Au2S CuF2 PbS SbF3 Work backwards - the overall charge in a compound must be 0 REVIEW CHAPTER 7 IF NEEDED AND COMPLETE # 40 ON PAGE 214 Read / review section 9.1 and complete #s 3-8 on page 269 Day 2 4-27 magnesium oxide Rb2S Fe2O3 calcium fluoride Rb2S NiO CuF Throwing in the transition metals: Lead II oxide Silver bromide Ferric Chloride cobalt III oxide cuprous chloride Beryllium sulfide iron III sulfide cuprous fluoride Au2S SrO AgF Anion Chemical bond Electrostatic force Cation Electronegativity Nonpolar covalent bond Binary ionic compound Octet rule Valence electrons Acids Ionic compound Transition metal Throwing in the transition metals: FeO Au2S CuF2 PbS SbF3 Work backwards - the overall charge in a compound must be 0 Throwing in the transition metals: Lead II oxide Silver bromide Ferric Chloride