Kristin Chin, Shivam Patel Madigan – P7 MP2 Project Molecular
advertisement

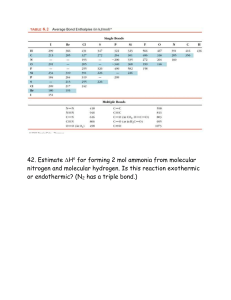
Kristin Chin, Shivam Patel MP2 Project Madigan – P7 Molecular Geometry and Lewis Structures Molecular Geometry and Lewis Structures Lewis Structure Key Terms: Valence electrons: electrons on the outermost shell of an atom - The Octet Rule: eight electrons in the valence shell (filling s and p orbitals) make an atom stable. Exceptions: Be - 2 bonds, B - 3 bonds, S - 6 bonds, P - 5 bonds, Xe – 6 bonds - Bond Lengths: Triple Bond < Double Bond < Single Bond - Bond Energy: Single Bond < Double Bond < Triple Bond - Resonance Structures: one or two of more Lewis structures for a single molecule can be drawn to represent a molecule. - Formal Charge: the difference between the number of valence electrons. The sum of the formal charges of the atoms in a molecule of ion must equal the charge on the molecule or ion. Equation: FC = total # of valence e- - total # of nonbonding e- - ½ (total # of bonding e-) Steps for Drawing Lewis Dot Structures: 1. Draw skeletal structure of compound showing what atoms are bonded to each other. Put least electronegative element in the center. 2. Count total number of valence electrons. Add 1 for each negative charge. Subtract 1 for each positive charge. 3. Complete an octet for all atoms except hydrogen 4. If structure contains too many electrons, form double and triple bonds on central atom as needed. Draw the Lewis structures and using the table on the back, predict the molecular geometry and label the bond angles for the following: 1. PO43- 2. CS2 2. SO42- 4. PH3 Kristin Chin, Shivam Patel MP2 Project Madigan – P7 Molecular Geometry and Lewis Structures VSEPR Theory: Shows how molecules are actually arranged in a 3D setting Predicting Molecular Geometry: 1. Draw Lewis structure for molecule PCl42. Count number of lone pairs on the central atom and number of atoms bonded to the central atom 3. Use VSEPR to predict the geometry of the molecule Number of Surrounding Atoms 2 2 2 2 2 3 3 3 Number of Lone Pairs Shape Bond Angle 0 1 2 3 4 0 1 2 Linear Bent Bent Linear Linear Trigonal Planar Trigonal Pyramidal T-Shaped 180 <120 104.5 180 180 120 107 90 and 180 3 3 T-Shaped 90 and 180 4 0 Tetrahedral 109.5 4 1 See-Saw <90 and <120 4 2 Square Planar 90 5 0 Trigonal bipyramidal 120 and 90 5 1 Square Pyramidal <90 6 0 Octahedral 90 Hybridization: mixing two or more atomic orbitals to form a new set of hybrid orbitals: # of lone pair on central atom + # of atoms bonded to central atom # of Bonded Atoms + # lone pairs 2 3 4 1. Determine the formal charge of : a. ClF3 b. OH3+ 2. Predict the hybridization of: a. CO2 b. CF Hybridization sp sp2 sp3 Examples SiO2 BF3 CCl4 3. Draw the Lewis structures and any resonance structures of: a. NO2b. HNO3 4. Name to molecular geometry of: a. XeOF4 b. SF5 Kristin Chin, Shivam Patel MP2 Project Madigan – P7 Molecular Geometry and Lewis Structures