Alice and Lewis Carroll
advertisement

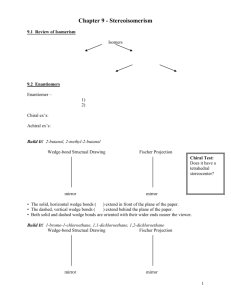
OPTICAL ISOMERISM OPTICAL ISOMERISM DEALS WITH CHIRALITY The word chiral derives from the Greek word ceir (cheir), meaning hand. Our hands are chiral - the right hand is a mirror image of the left as are most of life's molecules such as (R)-alanine and (S)-alanine, which are mirror images of each other. NOT ALL OBJECTS EXHIBIT CHIRALITY, BUT MOST OBJECTS** HAVE MIRROR IMAGES. EXCEPTIONS ARE: VAMPIRES,THE DEVIL, FALLEN MEN FAUST GOUNOD HOFFMAN (TALES OF HOFFMAN) HAD HIS REFLECTION STOLEN BY ANTONIO WHO GAVE IT TO DAPERTULU (A SORCEROR). Alone in his study, the aged Dr. Faust despairs that his lifelong search for a solution to the riddle of life has been in vain. Twice he raises a goblet of poison to his lips but falters when the songs of young men and women outside his window re-awaken the unfulfilled passions and desires of his youth. Cursing life and human passion, the envious philosopher calls on Satan for help. The Devil appears, and Faust tells him of his longing for youth and pleasure; Méphistophélès replies that these desires can be realized if he will forfeit his soul. Faust hesitates until the Devil conjures up a vision of a lovely maiden, Marguerite. A magic potion transforms Faust into a handsome youth, and he leaves with Méphistophélès in search of Marguerite (Duet: "A moi les plaisirs"). SO HOW ONE DISTINGUISHED MOLECULES THAT EXHIBIT CHIRALITY AND MOLECULES THAT LACK CHIRALITY (CALLED ACHIRAL MOLECULES)? MIRROR IMAGES OF CHIRAL MOLECULES ARE NON-SUPER POSABLE (THAT IS THEY ARE DIFFERENT) MIRROR IMAGES OF A CHIRAL MOLECULES ARE SUPER POSABLE (THAT IS THEY ARE THE SAME) CHIRAL OBJECTS: FEET, SCREWS, GOLF CLUBS, STUDENTS ARM CHAIRS, ENGLISH AND AMERICAN CARS ACHIRAL OBJECTS: SPOONS, KNIVES, FORKS Alice and Lewis Carroll Mathematic Don at Christ Church, Oxford University Real name was Charles Dodgson Amateur Photographer Alice’s father was Dean of Christ Church, Oxford WHAT DO CHIRAL MOLECULES LACK THAT ACHIRAL HAVE? CHIRAL MOLECULES LACK SYMMETRY A CHIRAL MOLECULES POSSESS SYMMETRY A symmetry element is a plane, a line or a point in or through an object, about which a rotation or reflection leaves the object in an orientation indistinguishable from the original. CONSIDER 1,2-DIMETHYLCYCLOPENTANE Me Me Me Me each has a plane of symmetry SAME, thus achiral Me Me Me Me different mirror images ASYMMETRIC CARBON ATOM (CHIRALITY CENTER) OR (STEREOCENTER) MOLECULE THAT HAS ONE OF THE ABOVE WILL BE CHIRAL THAT IS, LACKS SYMMETRY REQUIREMENT: 4 DIFFERENT GROUPS A A A D D C B B C D A B C B D C NOTE: EXCHANGE ANY TWO GROUPS TO GO FROM ENANTIOMER TO THE OTHER ENANIOMER!! EXAMPLES O Me H CHO NH2 OH H3C H OH H CH2OH O Cl OMe O O Me Me (R), (S) Nomenclature • Different molecules (enantiomers) must have different names. •Usually only one enantiomer will be biologically O •Configuration around the chiral carbon is specified with (R) and (S). C OH C H H3C NH2 natural alanine => Cahn-Ingold-Prelog Rules Overview Assign priorities to each group View molecule with the lowest priority away from the viewer Arrow is drawn from the atom with 1st priority through the atom with 2nd priority to the atom with 3rd priority. If arrow points clockwise - R(rectus) If arrow points counterclockwise - S (sinister) Assigning priorities-CASE 1 CASE 1 - four different atoms attached to chiral atom • Assign a priority number to each atom attached to the chiral carbon. •Atom with highest atomic number assigned the highest priorities #1. In case of isotopes, high(er)est priority given to the isotope with high(er)est mass number. 3-D Examples Assigning Priorities Cl 2 Cl 2 PERSPECTIVE 3 F H 4 Br 1 Br 1 2 Cl 1 Br 4 F 3 WEDGE 4 H Cl Cl Br H 3 Cl Br 1 F F 2 3 F H H 4 F Br H FISHER PRIORITY ASSIGNMENT EXAMPLES CASE 2 - In case of ties among 1st atom, go to 2nd atoms, then to 3rd etc until the tie is broken. Consider straight chain groups Me < Et<n-propyl etc Consider effect of branching Et< isopropyl < tert-butyl Consider effect of hetero atom Tert-butyl < CH2OMe Groups possessing multiple bonds C=C CC C-C Carbon doubly bonded to carbon is likened unto a carbon bonded to two carbons Assign Priorities O 2 OH C Cl 3 C 4 H H3C 1NH2 H natural alanine 3 H C H C 1 O CH2 *C 4 CH(CH ) CH2OH 2 expands to 3 2 4 H3 * 2 Cl 1 C C H C CH2 O *C CH(CH3)2 C CH2OH H O C => Assign (R) or (S) • Working in 3D, rotate molecule so that lowest priority group is in back. • Draw an arrow from highest to lowest priority group. • Clockwise = (R), Counterclockwise = (S) => Examples Fischer Projections • Flat drawing that represents a 3D molecule • A chiral carbon is at the intersection of horizontal and vertical lines. • Horizontal lines are forward, out-of-plane. • Vertical lines are behind the plane. Fischer Rules • Carbon chain is on the vertical line. • Highest oxidized carbon at top. • Rotation of 180 in plane doesn’t change molecule. • Do not rotate 90! • Do not turn over out of plane! => Fischer Mirror Images • Easy to draw, easy to find enantiomers, easy to find internal mirror planes. • Examples: CH3 CH3 CH3 H Cl Cl H H Cl Cl H H Cl H Cl CH3 CH3 CH3 => Fischer (R) and (S) • Lowest priority (usually H) comes forward, so assignment rules are backwards! • Clockwise 1-2-3 is (S) and counterclockwise 1-2-3 is (R). • Example: (S) CH3 (S) H Cl Cl H CH3 => Properties of Enantiomers • • • • Same boiling point, melting point, density Same refractive index Different direction of rotation in polarimeter Different interaction with other chiral molecules – Enzymes – Taste buds, scent => Optical Activity • Rotation of plane-polarized light • Enantiomers rotate light in opposite directions, but same number of degrees. => Polarimetry • • • • • Use monochromatic light, usually sodium D Movable polarizing filter to measure angle Clockwise = dextrorotatory = d or (+) Counterclockwise = levorotatory = l or (-) Not related to (R) and (S) => Biological Discrimination => Racemic Mixtures • • • • Equal quantities of d- and l- enantiomers. Notation: (d,l) or () No optical activity. The mixture may have different b.p. and m.p. from the enantiomers! => Racemic Products If optically inactive reagents combine to form a chiral molecule, a racemic mixture of enantiomers is formed. => Chirality of Conformers • If equilibrium exists between two chiral conformers, molecule is not chiral. • Judge chirality by looking at the most symmetrical conformer. • Cyclohexane can be considered to be planar, on average. => Mobile Conformers H H H H Br H Br Br H Br Nonsuperimposable mirror images, but equal energy and interconvertible. Br Br Use planar approximation. => Nonmobile Conformers If the conformer is sterically hindered, it may exist as enantiomers. => Allenes • Chiral compounds with no chiral carbon • Contains sp hybridized carbon with adjacent double bonds: -C=C=C• End carbons must have different groups. Allene is achiral. => Fischer Projections • Flat drawing that represents a 3D molecule • A chiral carbon is at the intersection of horizontal and vertical lines. • Horizontal lines are forward, out-of-plane. • Vertical lines are behind the plane. Fischer Rules • Carbon chain is on the vertical line. • Highest oxidized carbon at top. • Rotation of 180 in plane doesn’t change molecule. • Do not rotate 90! • Do not turn over out of plane! => Fischer Mirror Images • Easy to draw, easy to find enantiomers, easy to find internal mirror planes. • Examples: CH3 CH3 CH3 H Cl Cl H H Cl Cl H H Cl H Cl CH3 CH3 CH3 => Fischer (R) and (S) • Lowest priority (usually H) comes forward, so assignment rules are backwards! • Clockwise 1-2-3 is (S) and counterclockwise 1-2-3 is (R). • Example: (S) CH3 (S) H Cl Cl H CH3 => Diastereomers • Stereoisomers that are not mirror images. • Geometric isomers (cis-trans) • Molecules with 2 or more chiral carbons. => Alkenes Cis-trans isomers are not mirror images, so these are diastereomers. H H CH3 C C C C H3C H CH3 cis-2-butene H3C H trans-2-butene => Ring Compounds • Cis-trans isomers possible. • May also have enantiomers. • Example: trans-1,3-dimethylcylohexane CH3 CH3 H H H CH3 H CH3 => Two or More Chiral Carbons • Enantiomer? Diastereomer? Meso? Assign (R) or (S) to each chiral carbon. • Enantiomers have opposite configurations at each corresponding chiral carbon. • Diastereomers have some matching, some opposite configurations. • Meso compounds have internal mirror plane. • Maximum number is 2n, where n = the number of chiral carbons. => Examples COOH COOH H HO HO OH H H H OH COOH COOH (2S,3S)-tartaric acid (2R,3R)-tartaric acid COOH H OH H OH COOH (2R,3S)-tartaric acid => Fischer-Rosanoff Convention • Before 1951, only relative configurations could be known. • Sugars and amino acids with same relative configuration as (+)-glyceraldehyde were assigned D and same as (-)glyceraldehyde were assigned L. • With X-ray crystallography, now know absolute configurations: D is (R) and L is (S). • No relationship to dextro- or levorotatory. => D and L Assignments CHO H * CHO OH H CH2OH D-(+)-glyceraldehyde HO H COOH H2N * H => CH2CH2COOH L-(+)-glutamic acid OH H OH H * OH CH2OH D-(+)-glucose Properties of Diastereomers • Diastereomers have different physical properties: m.p., b.p. • They can be separated easily. • Enantiomers differ only in reaction with other chiral molecules and the direction in which polarized light is rotated. • Enantiomers are difficult to separate. => Resolution of Enantiomers React a racemic mixture with a chiral compound to form diastereomers, which can be separated. => Chromatographic Resolution of Enantiomers => End of Chapter 5