Periodic Table - 2i421scienceportfolio
advertisement

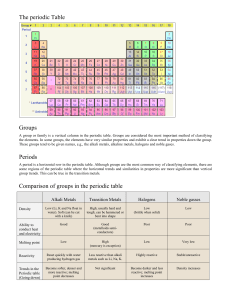
Periodic Table Lee Yun Jie 1i411 Tan Jing Ling 1i421 Jerry Yong 1i426 Periodic table: Groups of the periodic table: Although there are 8 columns in the periodic table, our project is focused on the group I, VII and 0. These groups are the Noble Gases, Alkali Metals, and Halogens How are elements in the periodic table grouped? The elements of the periodic table are grouped according to their characteristics. For example, the Alkali metals are known for their reactivity. Group 0: The Noble Gases The noble gases are a group of chemical elements with very similar properties: under standard conditions, they are all odourless, colourless, monatomic gases, with very low chemical reactivity. In fact, they are the least reactive elements in the Periodic Table. The six noble gases are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe) and radon (Rn). Characteristics All the noble gases have stable electronic arrangements. Except for helium, their atoms all have 8 electrons in their outer shells. That is why noble gases are so unreactive and do not usually form bonds with other atoms. The elements are all colourless gases. They consist of single atoms and are said to be monatomic. The elements have very low melting and boiling points. Uses Noble gases have several important applications in industries such as lighting, welding, and space exploration. Helium, neon and argon are the most widely used gases, because of their lack of reactivity. E.g.. Argon is used in light bulbs, neon is used in advertising strip lights, helium is used in modern airships. Group I: The Alkali Metals The Alkali Metals are the most reactive metals in the periodic table, volatile even with water. These metals are generally ionic a the have only one outer shell electrons. With reactivity increasing down the periodic table, these metals are Lithium, Sodium, Potassium, Rubidium, Caesium and Francium. Their generally reactive characteristics allows them to be found in several compounds. For example, the commonly found Sodium Chloride. Characteristics The Alkali metals are extremely reactive, with one outer shell electron, resulting in the formation of ions. Because of its reactivity with water, the Alkali Metals are never found as a pure substance, residing in several combinations of compounds. The densities of Alkali Metals increase down the periodic table. However, sodium is denser than potassium. With decreasing melting points as you move down the periodic table, Caesium and Francium are liquids at room temperature. Physical Features The Alkali Metals are shiny silvery solids which can be cut easily, although Caesium and Francium are liquids. These metals tarnish easily in air, therefore, to prevent a reaction, pure alkali metals are kept under oil. Alkali metals react vigorously with water, with the intensity of the reactions increasing as you move down the periodic table. Group VII: The Halogens The four halogens are chlorine, fluorine, bromine, iodine and astatine The artificially created element ununseptium or 117, named by the systematic name, might also be a halogen. The group of halogens is the only group of elements in the periodic table to contain elements in the three states of matter at room temperature Characteristics Halogens are naturally reactive, with Fluorine being the most highly reactive and they tend to react with the heavier noble gases. The melting and boiling points for halogens vary greatly, therefore, it is possible for all the halogens to have a different state of matter at room temperature. All halogens have 7 electrons in their outer shells, giving them a -1 oxidation number. Physical Features Solid halogens: Iodine & Astatine Liquid halogens: Bromine Gaseous halogens: Fluorine Uses Chlorine and Bromine are used as disinfectants in water and the process is known as bleaching. Iodine solution is used as a starch detector as in turns dark blue when it comes in contact with starch. Fluorine is added to toothpaste as it acts as a whitening agent to teeth. References: http://periodic.lanl.gov/what.htm http://en.wikipedia.org/wiki/Halogen http://www.halogens.com/ http://en.wikipedia.org/wiki/Periodic_table http://en.wikipedia.org/wiki/Noble_gas http://en.wikipedia.org/wiki/Alkali_metal