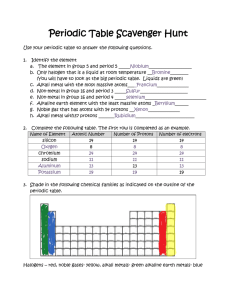
Unit B4– Photosynthesis
advertisement
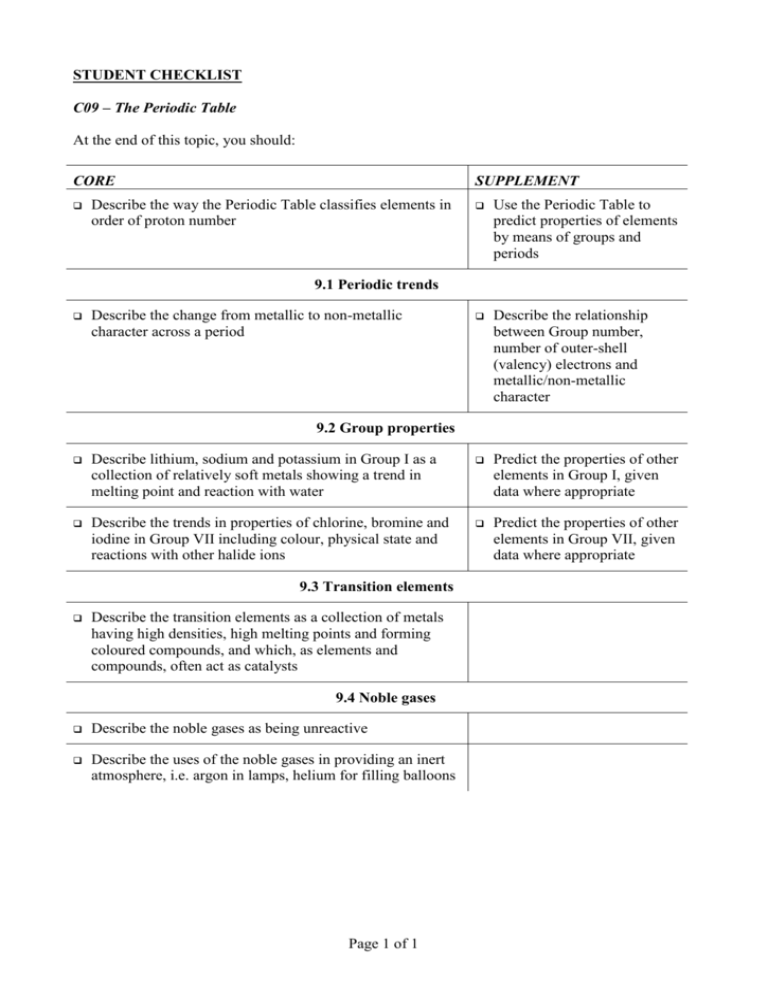
STUDENT CHECKLIST C09 – The Periodic Table At the end of this topic, you should: CORE SUPPLEMENT Describe the way the Periodic Table classifies elements in order of proton number Use the Periodic Table to predict properties of elements by means of groups and periods Describe the relationship between Group number, number of outer-shell (valency) electrons and metallic/non-metallic character 9.1 Periodic trends Describe the change from metallic to non-metallic character across a period 9.2 Group properties Describe lithium, sodium and potassium in Group I as a collection of relatively soft metals showing a trend in melting point and reaction with water Predict the properties of other elements in Group I, given data where appropriate Describe the trends in properties of chlorine, bromine and iodine in Group VII including colour, physical state and reactions with other halide ions Predict the properties of other elements in Group VII, given data where appropriate 9.3 Transition elements Describe the transition elements as a collection of metals having high densities, high melting points and forming coloured compounds, and which, as elements and compounds, often act as catalysts 9.4 Noble gases Describe the noble gases as being unreactive Describe the uses of the noble gases in providing an inert atmosphere, i.e. argon in lamps, helium for filling balloons Page 1 of 1