The Mole Concept - Grade10ScienceISZL
advertisement

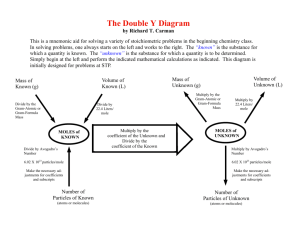
The Mole Concept The Mole Concept It is very difficult to deal with individual atoms in Chemistry because they are way too small, and reactions occur on the scale of millions of atoms rather than one or two. Therefore, chemists use the MOLE when talking about atoms. (The word mole is the English version of the German word mol, the shortened form of molekulargewicht meaning molecular weight) Using the term mole is similar to the way we use the term dozen. The word dozen stands for the number 12. One dozen eggs is the same as twelve eggs. The word mole stands for 6.02 X 1023. In one mole of an element, there are 6.02 X 1023 atoms of that element. The Mole Concept • The formal definition of the mole (abbreviated mol and given the symbol n) is, “The amount of substance which contains as many elementary particles as there are atoms in 12g of carbon12” • In 12g of carbon-12 there are exactly 6.02 X 1023 atoms of carbon. This number is known as Avogadro's constant and is given the symbol NA. The Mole Concept. Remember: MOLE is just a number - it stands for 6.02 X 1023 Celebrating the Mole • Between 6.02 am and 6.02 pm on the 23rd October is What a big number Avogadro’s number is extraordinarily large – If it were possible to make a mole of photocopy paper and pile the paper in a tower, the paper tower would stretch way past the Sun. A computer counting 10 billion times every second would take 2 million years to reach 6.02 X 1023 . 1 mole of marshmallows would cover Australia to a depth of 900 km. The Mole Concept. NA = 6.02 X 1023 Therefore, 1 mole is the same as 6.02 X 1023 particles. 1 mole H2O = 6.02 X 1023 molecules of water and contains 6.02 X 1023 atoms of oxygen and 2 x 6.02 X 1023 atoms of hydrogen or 12 x 1023 atoms of hydrogen or 1.2 x 1024 atoms of hydrogen. 1 mole of Na+ = 6.02 X 1023 ions of sodium Counting Particles We can use the formula below to help us count particles: Number of particles = number of mole x NA N = n x 6.02 x 1023 This formula can be rearranged to calculate the number of moles of a substance: Number of mole (n) = number of particles Avogadro’s number Complete the worksheet on The Mole concept: Counting Particles Molar Mass (M) • This is defined as the mass of 1 mol of the element expressed in grams per mole (g.mol-1) e.g. Molar mass of Cu atoms = mass of 1 mol of Cu atoms = 63.5 g.mol-1 (you get this number from the periodic table) M(Cu) = 63.5 g.mol-1 Therefore 63.5 g of Cu contains 6.02 x 1023 atoms of copper. • What is the molar mass of Fe atoms? • How many mole are there in 20.0g of calcium atoms? Molar Mass The molar mass of compounds can be found from the periodic table e.g. M(H2O) = 2 x 1.0 + 16 = 18.0 Therefore M(H2O) = 18 g.mol-1. What is the molar mass of CaCO3? What is the molar mass of glucose C6H12O6? Moles and Mass . Divide by molar mass Mass n=m M Multiply by molar mass. Moles where n = number of mol m = mass M = molar For example Determine the number of mole present in 32.0 g of oxygen atoms From the periodic table M(O) = 16.0g Therefore n(O) = 32.0 16.0 = 2.0 mol Moles and Mass . Divide by molar mass Mass n=m M Number of particles Moles Multiply by molar mass. where n = number of mol m = mass M = molar Divide by 6.02 x 1023 Multiply by 6.02 x 1023 Moles