Einstein's Photoelectric Law
advertisement

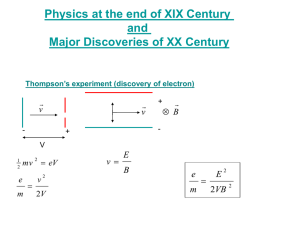
The Electron • Named by Stoney (Irish contribution ) • Indivisible quantity of charge measured by Millikan in the oil drop experiment • Negative • Orbits the nucleus • Very small mass Thermionic effect The emission of electrons from a hot metal. Photoelectric effect The emission of electrons due to electromagnetic radiation of the correct frequency. Photon A bundle of electromagnetic energy. Threshold frequency The lowest frequency that causes photo emission. Einstein’s photoelectric law mv hf = Φ + 12 2 Thermionic Emission • This is the release of electrons from the surface of a hot metal. • Cathode rays are beams of released electrons. The cathode ray tube-found in TVs and computer monitors The Photoelectric Effect The Photoelectric Effect This is the release of electrons from the surface of a metal due to electromagnetic radiation of suitable frequency. No electrons are released until the frequency increases above a certain threshold value. f0 To Demonstrate the photoelectric Effect • Attach a negatively charged zinc plate to an electroscope • Note that the leaves diverge. • Shine ultraviolet light on the zinc plate. • The leaves collapse due to the release of electrons caused by the photoelectric effect. (What happens if the electroscope is positively charged?) The Electromagnetic Spectrum The Photocell • Filters can be used to examine the effect of frequency on photocurrent. • The light intensity can be varied by changing the distance from the photocell to the light source. Effect of intensity and frequency As the frequency increases above the threshold, the energy of the electrons Current increases. As the light intensity increases the photocurrent increases. high intensity low intensity Frequency of incident light Einstein's explanation of the photoelectric effect • Light consists of bundles of electromagnetic energy called photons. • The energy of a photon depends on its frequency. E=hf where h=Plancks constant and f=frequency. • Each photon liberates one electron. • Increasing the intensity of the light increases the number of photons. The Work Function Φ=hf0 • • • • This is the energy needed to just free an electron but not give it any kinetic energy. This will take a minimum frequency called the threshold frequency. This threshold frequency is different for different metals. This is the frequency below which no electrons are released. Applications of the photoelectric effect Solar panels Automatic doors The Threshold Frequency This is the frequency below which no electrons are released. We use the same colour of light, the same target metal, but we use a brighter and brighter light. What is the difference A more intense light between lightmore of low sourceahas intensity, and one of photons.. high intensity? More photons free more electrons from Why does a more the metal. intense light cause a larger current to flow? Increased intensity frees more they How electrons come the but stopping have all theissame energy voltage the same aseven the frequency of the when the light is light brighter? is the same. Einstein's Photoelectric Law E mv 1 2 2 E mv 1 2 1 2 2 E = the energy of the photon =hf = the work function = hf0 2 mv = the kinetic energy of the electron Xrays • Discovered by Wilhelm Rontgen 1895. • They are high frequency electromagnetic radiation. • They cannot be deflected by electric or magnetic fields. • They can penetrate matter. • They ionise matter. • They cause fluorescence Uses of X-rays • Destroy cancerous cells. • Detect cracks in pipes. • Determining thickness of materials. How X-rays are produced. • • • • A heated filament releases electrons from the cathode by thermionic emission. A cooling system removes unwanted heat from the target. If the filament current is increased, more heat is released, therefore more electrons and as a result more x-rays are released. (intensity) If the voltage between cathode and anode is increased, the electrons are accelerated to higher speeds and so a higher energy photon or a more penetrating x-ray is produced. Why is X-ray production often described as the inverse of the photo electric effect? X-rays which are electromagnetic radiation are produced when high energy electrons strike a metal target whereas the photoelectric effect is the release of electrons by electromagnetic radiation.