Mole
advertisement

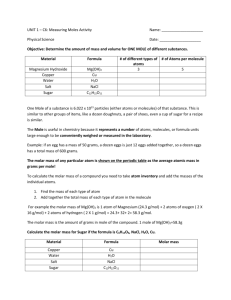
Mole Atomic mass: Mole: Molar Mass: Avogadro’s number Vocabulary: Atomic mass • the mass of an atom expressed in atomic mass units. Mole: • the amount of substance whose number of particles is the same as the number of atoms in 12 g of carbon-12 Molar Mass: • the mass in a gram of 1 mole of a substance Avogadro’s number • 6.022 x 10^ 23, number of atoms or molecules in a mole. The Mole Number of atoms in exactly 12 grams of atoms 12C How many atoms in 1 mole of 12C ? Based on experimental evidence 1 mole of 12C = 6.022 × 1023 atoms = 12.011 g Avogadro’s number = NA Number of atoms, molecules or particles in one mole 1 mole of X = 6.022 × 1023 units of X 1 mole Xe = 6.022×1023 Xe atoms 1 mole NO2 = 6.022×1023 NO2 molecules Jespersen/Brady/Hyslop Chemistry: The Molecular Nature of Matter, 6E 3 Finding Molar Mass Molar mass (also called “molecular weight” or “molecular mass”): The weight of one mole of a chemical compound. The unit is “g/mol”. For elements, the mass of one mole of atoms is called the “atomic mass” and is found on the periodic table. How to calculate the molar mass of a compound: For elements, the molar mass is the same thing as the atomic mass. For chemical compounds, it’s the sum of the masses of all of the atoms in the molecule. Example: NaCl Na: Cl: 23 grams x 1 atom = 35 grams x 1 atom = Total: 23 grams/mol 35 grams/mol 58 grams/mol More examples: o o o MgCl2 Fe(OH)2 Be3(PO4)2 94 g/mol 90 g/mol 217 g/mol For these last two examples, tell them that the molar mass for compounds like this is found by multiplying everything in the parentheses by the number outside the parentheses. Mole calculations: 1 mole of carbon atoms = 6.022x1023 atoms of carbon = weighs 12 grams 1 mole of oxygen molecules = 6.022x1023 molecules of oxygen = weighs 32 grams 1 mole of NO2 particles= 6.022x1023 particles of NO2 = weighs 30 grams 1 mole of fluoride ion= 23 ions of 6.022x10 fluoride ions = weighs 19 grams