The Grignard Reaction: Workup April 5 & 7, 2010
advertisement

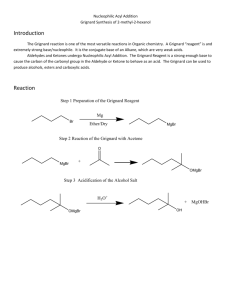
The Grignard Synthesis II Miniscale Synthesis of Triphenylmethanol from Ethyl Benzoate, Part 2 MgBr Organic Chemistry Lab II, Spring 2010 Dr. Milkevitch April 5 & 7, 2010 Today’s Experiment Complete last week’s experiment – Grignard synthesis React the Grignard with an ester – Make an alcohol Last Week Prepared the Grignard reagent Reacted it with ethyl benzoate – In ether Allowed it to sit until this week This week: – Workup Isolation & purification Characterization Procedure: Part 2 Retrieve your RB flask from the hood Take a look at it – Replace any lost volume with more ether Remove the stir bar Pure the reaction mixture into a 50 ml erlenmeyer flask that contains 8 g of ice Rinse the reaction flask with ~2 ml of ether and add this rinse to the erlenmeyer with the ice While stirring, carefully add 3M HCl dropwise to the solution – Use a glass stir rod to break up the solid Continue to add acid until the lower layer is just acidic to litmus – Blue litmus turns red if the solution is acidic Carefully pour the reaction mixture into your separatory funnel – Leave any unreacted magnesium in the flask Procedure, con’t Rinse the flask with 5 ml of diethyl ether – Add this to the separatory funnel Reaction mixture should separate into 2 layers Product is in the upper layer (ether layer) Separate the layers – Into separate erlenmeyer flasks 1)One will have ether layer 2)One will have the water layer – Put the water layer back into the separatory funnel Extract the water layer with 5 ml of diethyl ether – Allow layers to separate – Drain off the water layer into an erlenmeyer flask Drain off the ether wash, add it to the original ether wash (#1 above) Now, put all the ether layers into the separatory funnel – Extract it with 5 ml of DI H2O Procedure, con’t Remove the water – Store in a beaker, set aside Transfer the ether to a clean erlenmeyer flask – Dry over magnesium sulfate ~10 min Decant or gravity filter the dried solution – Into a pre-weighed 50 ml erlenmeyer flask Add a stir bar/boiling chip Evaporate off ether using a hot plate – In the hood Should get an off-white solid – Mixture of biphenyl and triphenylmethanol Add 10 ml of petroleum ether – Stir thoroughly – Vacuum filter with a Buchner funnel Weigh product Characterization Do a TLC – Plates are in the oven – Standards are in the hood Standards: Triphenyl methanol, biphenyl Spot these Also spot your compound! Develop your plate in methylene chloride Visualize under UV light Submit a sample for GC analysis Dissolve a small amount of your compound in methylene chloride Place in a small vial – Vials are in the hood – Label with your names Your Report Formal lab report not required Complete experiment worksheet