2.Acidity and Alkalinity
advertisement

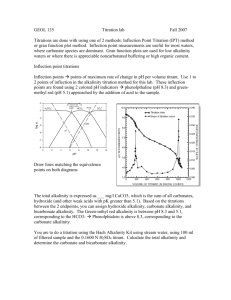
Acidity and Alkalinity Definitions Alkalinity Measurement ----------- Task ----------Acidity Measurement Importance What are Acidity and Alkalinity Acidity The capacity of a water sample to neutralise added alkali. Two Types Mineral Acidity from Strong Acids, and from Salts of Trivalent Metal Ions H2SO4 2 H+ + SO42FeCl3 + 3 H2O Fe(OH)3 + 3 H+ + 3 Cl - Carbon Dioxide Acidity (Weak Acidity) CO2 + H2O H2CO3* H+ + HCO3- What are Acidity and Alkalinity Alkalinity The capacity of a water sample to neutralise added acid. Mainly from: 1. The Carbonate - Bicarbonate buffering system. 2. The salts of weak acids CO2 + CaCO3 + H2O Na Acetate, Na Propionate 3. Hydroxide Ammonia Ca2+ + HCO3- Equilibrium of CO2 , HCO3- , CO32CO2 (g) 350ppm air water CO2 (aq) + H2O H2CO3 10-5M pH H+ + HCO3- pKa = 6.3 4 4.5 5 6 pH 7 2H+ + CO32pKa = 10.3 8 8.3 9 10 = pKa + log ([A-] / [AH]) When reaction is 99% to the left then pH = 6.3 + log (1/100) = 6.3 - 2 pH = 4.3 How is Acidity Determined By titration with a Standard Alkali solution to a specific Endpoint. Mineral Acidity Indicator used is Methyl Orange (endpoint at pH 4.5 ) Carbon Dioxide Acidity Indicator used is Phenolphthalein (endpoint at pH 8.3 ) In practice CO2 is the major weak acid. Others are possible, eg. Acetic Acidity High P endpoint (pH 8.3) P pH starting pH M endpoint (pH 4.5) M Low starting pH M = Methyl Orange P = Phenolphthalein Strong Acids Weak Acids (CO2 Acidity) Titration with N/50 NaOH How is the Acidity Calculated and Reported Titration Volume, Alkali Strength, Sample Volume Usually Report Acidity as Calcium Carbonate Equivalent “ The Acidity to pH X or = Y mg CaCO3 / litre “ Methyl Orange Acidity / Mineral Acidity = Phenolphthalein Acidity / CO2 Acidity = If N/50 NaOH is used Acidity (mg CaCO3) = Vt x 1000/ Vs Vt = Titre volume (ml) Vs = Sample volume (ml) How is Alkalinity Determined By titration with a Standard Acid solution to a specific Endpoint. Sample pH Values > pH 8.3 Two Step Titration Step 1. Indicator used is Phenolphthalein (endpoint at pH 8.3 )* Step 2. Indicator used is Methyl Orange (endpoint at pH 4.5 )** Step 1. neutralises alkalinity due to strong bases (OH- , CO32-) Step2. neutralises alkalinity due to weaker bases (HCO3-) * chosen as CO32- HCO3- reaction is complete. ** chosen as HCO3- H2CO3 reaction is complete. How is Alkalinity Determined Sample pH Values < pH 8.3 One Step Titration Step 1. Indicator used is Methyl Orange (endpoint at pH 4.5 ) only Bicarbonate present when pH < 8.3 at start Interpretation of Data When Sample pH value is > pH 8.3 a number of base species can potentially exist together. 1. 2. 3. 4. Carbonate and Bicarbonate Carbonate only Hydroxide and Carbonate Hydroxide only pH > 8.3 pH > 10 pH > 10 pH >>11 Alkalinity (5 cases) High P>1/2M P=1/2M starting pH A P Titration with N/50 H2SO4 P<1/2M B M starting pH X P endpoint (pH 8.3) pH X B Y M endpoint (pH 4.5) Low M = Methyl Orange P = Phenolphthalein Hydroxide Carbonate Bicarbonate P=a+b M=b+P P=x M = 2x + y Hydroxide = a = 2P - M Carbonate = 2b = 2M - 2P Carbonate = 2x = 2P Bicarbonate = y = M - 2P How is the Alkalinity Calculated and Reported Titration Volume, Acid Strength, Sample Volume Usually Report Alkalinity as Calcium Carbonate Equivalent ie. For each of the 3 Classes mg CaCO3 / litre Hydroxide Alkalinity Carbonate Alkalinity Bicarbonate Alkalinity If N/50 H2SO4 is used Alkalinity (mg CaCO3) = Vt x 1000/ Vs Vt = Titre volume (ml) Vs = Sample volume (ml) Application of Acidity and Alkalinity Data Acidity Selection of new Water Supplies minimise treatment costs (Lime, NaOH) Industrial Wastewater Metal Pickling Liquor (phosphoric acid) Consent to Discharge Alkalinity Calculate safe levels of Ferric and Alum Biological WWT Plants - gives buffering capacity Potable - range 50 - 300 mg/l Industrial - consent to discharge